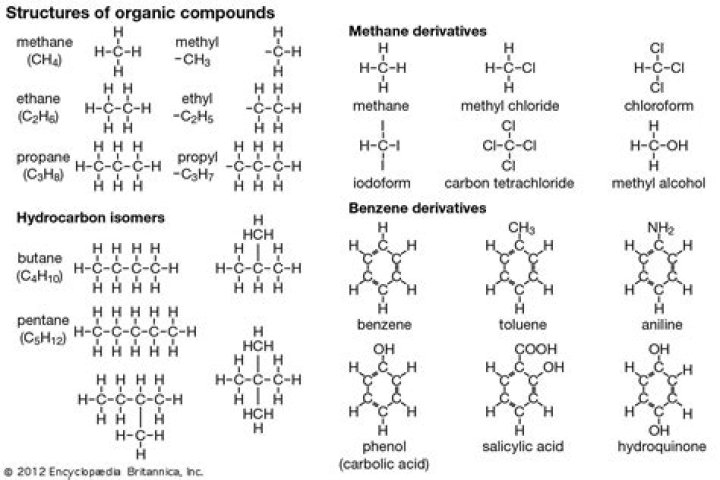
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons from which one hydrogen atom has been removed are functional groups called hydrocarbyls..
Likewise, what are examples of hydrocarbons?
Compounds like methane, butane, propane, and hexane are all hydrocarbons. Their chemical formulas consist of only carbon and hydrogen atoms, in a variety of ratios and chemical configurations. 2.
Subsequently, question is, what are 5 common hydrocarbons? Common hydrocarbons:
- Methane(CH4)
- Ethane(C2H6)
- Propane(C3H8)
- Butane(C4H10)
- Pentane(C5H12)
- Hexane(C6H14)
Additionally, what is hydrocarbon used for?
Uses of Hydrocarbons The most important use of hydrocarbons is for fuel. Gasoline, natural gas, fuel oil, diesel fuel, jet fuel, coal, kerosene, and propane are just some of the commonly used hydrocarbon fuels. Hydrocarbons are also used to make things, including plastics and synthetic fabrics such as polyester.
What is hydrocarbon products?
Hydrocarbon. Hydrocarbons are compounds comprised exclusively of carbon and hydrogen and they are by far the dominant components of crude oil, processed petroleum hydrocarbons (gasoline, diesel, kerosene, fuel oil, and lubricating oil), coal tar, creosote, dyestuff, and pyrolysis waste products.
Related Question Answers
Is water a hydrocarbon?
A hydrocarbon is an organic chemical compound composed exclusively of hydrogen and carbon atoms. Hydrocarbons occur naturally and form the basis of crude oil, natural gas, coal, and other important energy sources. Hydrocarbons are highly combustible, producing carbon dioxide, water, and heat when burnt.Is sugar a hydrocarbon?
The major differences between sugars and hydrocarbons are: Solubility: Sugars are very polar, due to their -OH groups, and dissolve readily in water by forming many hydrogen bonds. Energy content: Sugars contain only half as much energy as hydrocarbons.Which hydrocarbon is most reactive?
alkynes
Is wood a hydrocarbon?
Seasoned wood (wood that has been allowed to sit for a year or two) or kiln-dried wood contains a lot less water, but it still contains some. These compounds are all combustible (gasoline and alcohol are, after all, hydrocarbons -- the volatile hydrocarbons in wood burn the same way). Carbon.Is co2 a hydrocarbon?
Common properties of hydrocarbons are the facts that they produce steam, carbon dioxide and heat during combustion and that oxygen is required for combustion to take place. The simplest hydrocarbon, methane, burns as follows: Burning of hydrocarbons is an example of an exothermic chemical reaction.Is coal a hydrocarbon?
Hydrocarbon resource. Hydrocarbon resources are resources that contain hydrocarbon molecules which means it consists both hydrogen and carbon. Hydrocarbon resources are often known as fossil fuels (natural gas, oil, and coal) since hydrocarbons are the primary constituent in these.Is petrol a hydrocarbon?
Petroleum is a mixture of a very large number of different hydrocarbons; the most commonly found molecules are alkanes (paraffins), cycloalkanes (naphthenes), aromatic hydrocarbons, or more complicated chemicals like asphaltenes. Alkanes with more than 16 carbon atoms can be refined into fuel oil and lubricating oil.Is c3h8 a hydrocarbon?
Uses: A colorless, easily liquefied, gaseous hydrocarbon (compound of carbon and hydrogen), the third member of the paraffin series following methane and ethane. The chemical formula for propane is C3H8. From propylene such organic chemicals as acetone and propylene glycol are derived.Is Hydrocarbon harmful to humans?
Hydrocarbons are oily liquids. Many are not harmful unless the oily liquid gets into the lungs. However, if it enters the lungs, it can cause a pneumonia-like condition; irreversible, permanent lung damage; and even death.How is hydrocarbon produced?
Hydrocarbon gas liquids are derived from natural gas and crude oil. Hydrocarbon gas liquids (HGL) are produced when raw natural gas is processed at natural gas processing plants and when crude oil is refined into petroleum products.How is hydrocarbon made?
Hydrocarbons are organic compounds that are made of only hydrogen and carbon atoms. They are found in many places, including crude oil and natural gas. Learn the different forms of these simple, yet varied, organic compounds.Which hydrocarbon is used as a fuel?
The greatest amounts of hydrocarbons are used as fuel for combustion, particularly in heating and motor fuel applications. The primary components of natural gas are methane and ethane.Is wax a hydrocarbon?
All waxes are essentially hydrocarbons, which means they are largely composed of hydrogen (H) and carbon (C) atoms. When you light a candle, the heat of the flame melts the wax near the wick. This liquid wax is then drawn up the wick by capillary action.When was hydrocarbon discovered?
Hydrocarbon exploration began in 1936 and has resulted in the discovery of about 200 oil and gas accumulations. Most of these are contained in Carboniferous to Permian series that were deposited during the post-rift stage of the basin evolution (Fig.Can a hydrocarbon contain oxygen?
Aliphatic hydrocarbons are primarily composed of hydrogens and carbons. However, they may contain oxygen, nitrogen, sulfur, and chlorine. Aliphatic hydrocarbons may be saturated or unsaturated. Saturated compounds are made only of single bonds and contain the maximum number of hydrogens.What are the properties of hydrocarbons?
Hydrocarbons are organic compounds composed of only carbon and hydrogen. The alkanes are saturated hydrocarbons—that is, hydrocarbons that contain only single bonds. Alkenes contain one or more carbon-carbon double bonds. Alkynes contain one or more carbon-carbon triple bonds.What are the major sources of hydrocarbons?
The natural sources of hydrocarbons include coal, petroleum, and natural gas.Is benzene a hydrocarbon?
Benzene is an organic chemical compound with the chemical formula C6H6. The benzene molecule is composed of six carbon atoms joined in a ring with one hydrogen atom attached to each. As it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon.Is hydrocarbon bad for the environment?
There are various impacts of combustion on the environment, these impacts can be caused by; Gas leaks, oil spillage, noise and air pollution. Incomplete combustion of hydrocarbons also results in carbon monoxide pollution. An odorless, colorless gas, carbon monoxide can be harmful to both the environment and to people.