How the body regulates pH balance?
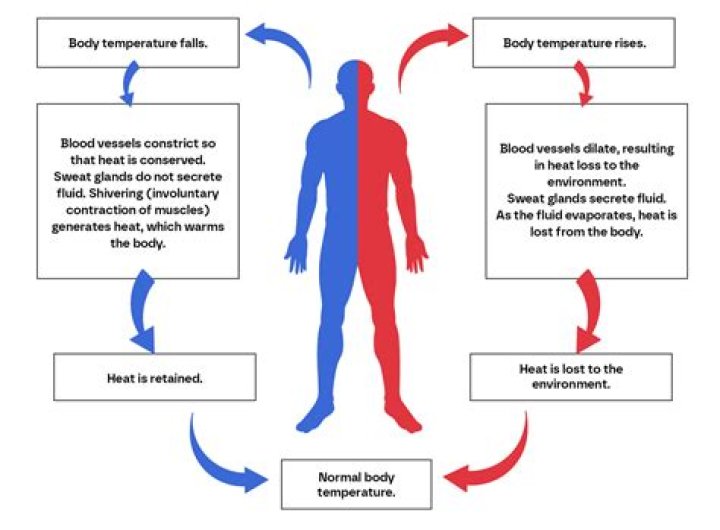
.
Beside this, how acid base balance is maintained in the body?
The kidneys help control acid-base balance by excreting hydrogen ions and generating bicarbonate that helps maintain blood plasma pH within a normal range. Protein buffer systems work predominantly inside cells.
Also Know, what happens if pH is too high in body? When your body fluids contain too much acid, it's known as acidosis. Acidosis occurs when your kidneys and lungs can't keep your body's pH in balance. A lower pH means that your blood is more acidic, while a higher pH means that your blood is more basic.
Also asked, what are the three major mechanisms of pH regulation?
There are three mechanisms which diminish pH changes in body fluid: buffers; respiratory; renal. (a) Proteins are the most important buffers in the body. They are mainly intracellular and include haemoglobin.
How does the kidney regulate pH?
The kidneys can regulate reabsorption of carbonic acid in the tubule, increasing or reducing acid secretion. So, urine that is more acidic than normal may mean the body is ridding itself of excess dietary acid and thus making blood pH more alkaline. Ammonia is another way the kidney can regulate pH balance.
Related Question AnswersHow do you know if your body is alkaline?
First, a little chemistry: A pH level measures how acid or alkaline something is. A pH of 0 is totally acidic, while a pH of 14 is completely alkaline. Your blood is slightly alkaline, with a pH between 7.35 and 7.45. Your stomach is very acidic, with a pH of 3.5 or below, so it can break down food.How is pH maintained in the body?
Buffers in the Human Body Blood contains large amounts of carbonic acid, a weak acid, and bicarbonate, a base. Together they help maintain the bloods pH at 7.4. The bicarbonate neutralizes excess acids in the blood while the carbonic acid neutralizes excess bases.What are the symptoms of too much alkaline in the body?
Too much alkalinity may also agitate the body's normal pH, leading to metabolic alkalosis, a condition that may produce the following symptoms:- nausea.
- vomiting.
- hand tremors.
- muscle twitching.
- tingling in the extremities or face.
- confusion.
Does your body regulate pH levels?
The lungs control your body's pH balance by releasing carbon dioxide. Your brain constantly monitors this in order to maintain the proper pH balance in your body. The kidneys help the lungs maintain acid-base balance by excreting acids or bases into the blood.How do you remove acid from your body?
Eat vegetables such as spinach, broccoli and beans or fruits such as raisins, bananas and apples are appropriate choices for neutralizing body pH. 5. Acid imbalance can result from poorly controlled diabetes which is treated with insulin to prevent metabolic acidosis.What is normal pH level?
A pH of 7 is neutral. The lower the pH, the more acidic the blood. A variety of factors affect blood pH including what is ingested, vomiting, diarrhea, lung function, endocrine function, kidney function, and urinary tract infection. The normal blood pH is tightly regulated between 7.35 and 7.45.What are the 3 buffer systems in the body?
There are several buffer systems in the body. The most important include: (1) bicarbonate buffer (HCO3–/CO2), (2) haemoglobin buffer (in erythrocytes), (3) phosphate buffer, (4) proteins, and (5) ammonium buffer.What is the pH of urine?
According to the American Association for Clinical Chemistry, the average value for urine pH is 6.0, but it can range from 4.5 to 8.0. Urine under 5.0 is acidic, and urine higher than 8.0 is alkaline, or basic. Different laboratories may have different ranges for "normal" pH levels.How is pH regulated?
The kidneys have the predominant role in regulating the systemic bicarbonate concentration and hence, the metabolic component of acid-base balance. Normally, systemic acid-base balance is well regulated with arterial pH between 7.36 and 7.44; intracellular pH is usually approximately 7.2.What is the pH value of water?
pH and Water The pH of pure water is 7. In general, water with a pH lower than 7 is considered acidic, and with a pH greater than 7 is considered basic. The normal range for pH in surface water systems is 6.5 to 8.5, and the pH range for groundwater systems is between 6 to 8.5.How do buffers resist changes in pH?
Buffers are solutions that resist changes in pH, upon addition of small amounts of acid or base. The can do this because they contain an acidic component, HA, to neutralize OH- ions, and a basic component, A-, to neutralize H+ ions. Since Ka is a constant, the [H+] will depend directly on the ratio of [HA]/[A-].What system regulates pH in the body?
Acid–base balance. The pH of the extracellular fluid, including the blood plasma, is normally tightly regulated between 7.32 and 7.42, by the chemical buffers, the respiratory system, and the renal system.What can change the pH of blood?
Fluid loss Losing too much water from your body can increase blood pH. This happens because you also lose some blood electrolytes — salts and minerals — with water loss. These include sodium and potassium.How does pH affect the nervous system?
The normal pH of the body is 7.4. It is therefore vital that it can sense pH levels. Inhibitory effect. When the nerve system is active, it releases lactic acid metabolically, and at high levels of activity, such as during intense exercise or epilepsy, this causes the pH in the nerve system to become more acidic.How do I calculate pH?
To calculate the pH of an aqueous solution you need to know the concentration of the hydronium ion in moles per liter (molarity). The pH is then calculated using the expression: pH = - log [H3O+].Where is bicarbonate produced in the body?
Bicarbonate also acts to regulate pH in the small intestine. It is released from the pancreas in response to the hormone secretin to neutralize the acidic chyme entering the duodenum from the stomach.How do animals regulate blood pH?
The buffering systems that regulate blood pH in mammals are most likely the same in reptiles; the bicarbonate–carbonic acid buffer system is the most important because of the rapid rate of CO2 elimination via the lungs after conversion from H2CO3.What are the signs of an unbalanced pH?
Signs and symptoms of an unbalanced vaginal pH- a foul or fishy smell.
- unusual white, gray, or green discharge.
- vaginal itching.
- burning when you urinate.
What foods are high in acid?
High-acid food and drink- grains.
- sugar.
- certain dairy products.
- fish.
- processed foods.
- fresh meats and processed meats, such as corned beef and turkey.
- sodas and other sweetened beverages.
- high-protein foods and supplements.