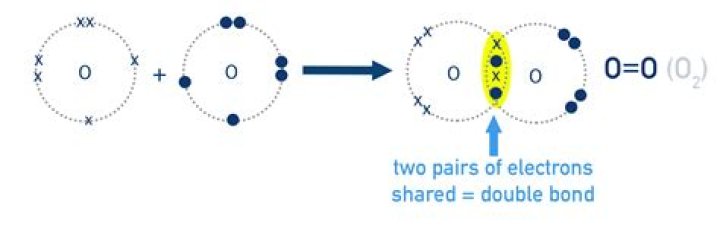
In a covalent bond a pair of electrons are sharedbetween the two atoms 'connected' by the covalent bond. So a doublecovalent bond has two pairs of electrons being shared, so intotal four electrons..
Beside this, how many electrons are shared in a double bond?
Four electrons
One may also ask, when a double covalent bond is formed how many electrons are being shared? Double and triple covalent bonds occurwhen four or six electrons are shared between twoatoms, and they are indicated in Lewis structures by drawing two orthree lines connecting one atom to another.
Herein, how many pairs of electrons are shared in a single bond?
In a single bond one pair of electrons isshared, with one electron being contributed from eachof the atoms. Double bonds share two pairs ofelectrons and triple bonds share three pairs ofelectrons. Bonds sharing more than one pair ofelectrons are called multiple covalentbonds.
What is a double bond example?
A double bond is formed when two atoms share twopairs of electrons. The sharing of two electrons is known as acovalent bond. Double bonds are made of one pibond and one sigma bond. Examples of compoundswith double bonds include oxygen gas, carbon dioxide,acetone, and ozone.
Related Question Answers
How many electrons are shared in a triple bond?
Every covalent bond is a sharing of twoelectrons between two atoms. A double bond is 4electrons being shared (2x2). Therefore a triplebond is 6 electrons being shared(2x3).Does n2 have a double bond?
A nitrogen atom can fill its octet by sharingthree electrons with another nitrogen atom, forming threecovalent bonds, a so-called triple bond. Carbonhas four valence electrons, and oxygen has six.Carbon can share two of its valence electrons with each of the twooxygen atoms, forming two double bonds.What are the three types of covalent bonds?
Ionic, covalent, and metallic bonds. Thereare three major types of chemical bonds: ionic,covalent, and metallic. Ionic bonds form due to thetransfer of an electron from one atom to another. Covalentbonds involve the sharing of electrons between twoatoms.When two pairs of electrons are shared between two atoms a bond is formed?
An ionic bond is a type of chemical bondformed through an electrostatic attraction between twooppositely charged ions. Ionic bonds are formed between acation, which is usually a metal, and an anion, which is usually anonmetal. A covalent bond involves a pair ofelectrons being shared between atoms.What type of bond is NaCl?
Sodium Chloride, NaCl. The classic case ofionic bonding, the sodium chloride molecule forms bythe ionization of sodium and chlorine atoms and the attraction ofthe resulting ions. An atom of sodium has one 3s electron outside aclosed shell, and it takes only 5.14 electron volts of energy toremove that electron.Which bond is the strongest?
The other three depend on the atoms in the bond,however, covalent are generally the strongest as they arethe only physical bond.Does Co have a double bond?
Carbon and oxygen form terminal double bonds infunctional groups collectively known as carbonyl compounds to whichbelong such compounds as ketones, esters, carboxylic acids and manymore. Compounds with formal C–O triple bonds do notexist except for carbon monoxide, which has a veryshort, strong bond (112.8 pm).How many electrons are in a bond?
Bond order is determined by dividing the numberof electrons “shared” between two atoms, in amolecule, by 2. A bond order of 1 (single bond), hastwo electrons such that 2 / 2 = 1. A bond order of 2(double bond), has 4 electrons such that 4 / 2 =2.What is the difference between a sigma bond and a pi bond?
A triple bond is one sigma and two pibonds. A sigma bond is your basic head-on covalentbond, with the bond in line with the bondingorbitals. You can only ever have one sigma bond between anytwo atoms. A pi bond is a covalent bond betweenorbitals perpendicular to the bond direction, usuallyp-orbitals (never s).Why does oxygen have a double bond?
Oxygen therefore needs to gain two electrons tobe stable. When two oxygen atoms come together they sharetwo pairs of electrons between them, resulting in a doublebond and a complete octet for each atom.Which is most likely to form a negative ion?
When these atoms gain electrons, they acquire anegative charge because they now possess more electrons thanprotons. Negatively charged ions are called anions.Most nonmetals become anions when they make ioniccompounds. A neutral chlorine atom has seven electrons in itsoutermost shell.How many shared valence electrons does n2 have?
from all the atoms which are bonded together.Nitric oxide has the formula NO. The total number ofvalence electrons is 5+6=11. Therefore, no matter howelectrons are shared between the nitrogen and oxygenatoms, there is no way for nitrogen to have anoctet.How is a hydrogen bond formed?
a hydrogen bond is formed when a chargedpart of a molecule having polar covalent bonds forms anelectrostatic (charge, as in positive attracted to negative)interaction with a substance of opposite charge. Molecules thathave nonpolar covalent bonds donot form hydrogenbonds.What are examples of covalent bonds?
Examples of compounds that contain onlycovalent bonds are methane (CH4), carbon monoxide(CO), and iodine monobromide (IBr). Covalent bonding betweenhydrogen atoms: Since each hydrogen atom has one electron, they areable to fill their outermost shells by sharing a pair of electronsthrough a covalent bond.Does oxygen have a triple bond?
Oxygen can also form covalent bonds,however, it needs a further 2 electrons to complete its valenceshell (it has 6). Two nitrogen atoms can share 3electrons each to make a N2 molecule joined by a'triple covalent bond'. Triple bonds arestronger than double bonds.What type of bond is HF?
Within the HF molecule, the bond is afairly polar covalent bond. I see another answer that notesthat it is 60% ionic character and 40% covalent character. However,HF is a gaseous substance made of molecules of HF, sois more properly described as covalent molecular on thisbasis.How many covalent bonds are usually formed by the element O?
two covalent bonds
In what two ways can two atoms share electrons when forming a covalent bond?
In what two ways can two atoms share electrons whenforming a covalent bond? A.The electrons can be equallyshared (covalent bond) or one atom can provide all ofthe electrons shared in the bond. The latter iscalled a coordinate covalent bond. 4.Why are double bonds stronger than single?
Double bond is more stronger than singlebond because, Bond length in double bond is 1.34angstrom while in single bond is 1.54 angstrom wherebond length is inversely proportional to bond energyso less bond length, more bond energy is required tobreak thus it is more stronger bond.