Will isopropyl alcohol evaporate in the freezer?
.
Herein, can isopropyl alcohol evaporate?
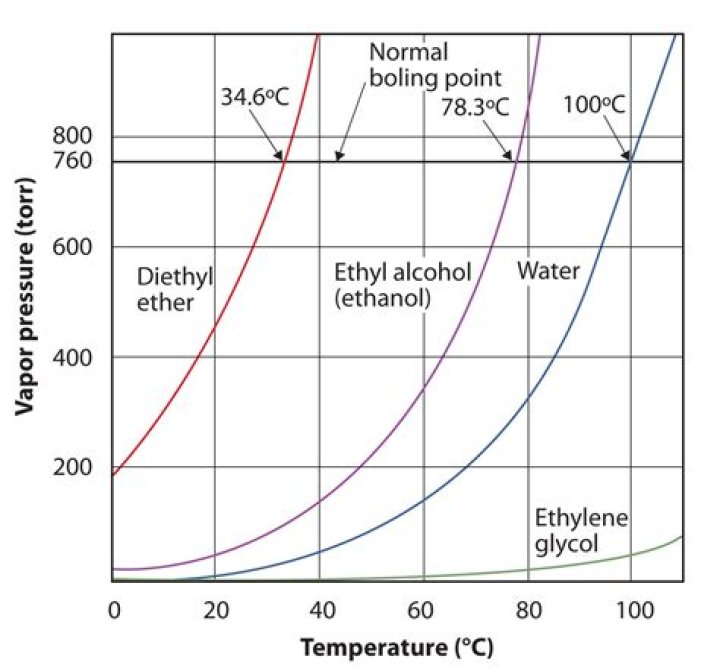
Pure isopropyl alcohol would normally evaporate completely at room temperature in our standard atmosphere. Like other volatile liquids, it evaporates because the molecules have enough kinetic energy at room temperature to overcome the forces (attractions) that otherwise hold the molecules in liquid form.
Likewise, will rubbing alcohol evaporate in the cold? Rubbing alcohol feels even colder, however, because about 70% of it is made up of isopropyl alcohol. The molecules of isopropyl alcohol don't stick together as strongly at room temperature as water molecules do, which means the alcohol evaporates more quickly than water does.
Keeping this in view, can you freeze isopropyl alcohol?
Rubbing alcohol has a freezing point between -26 and -58 degrees F, depending on the percentage of isopropyl alcohol to water, making it virtually impossible to freeze in a home freezer. More alcohol will make the ice pack slushier.
Is isopropyl alcohol flammable after it dries?
Isopropyl alcohol is highly flammable and can easily ignite. Isopropyl alcohol should be kept away from heat, sparks, flames and other sources of ignition, as well as strong oxidizers, acetaldehyde, chlorine, ethylene oxide, acids, and isocyanates.
Related Question AnswersDoes isopropyl alcohol evaporate quickly?
Isopropyl alcohol dissolves a wide range of non-polar compounds. It also evaporates quickly, leaves nearly zero oil traces, compared to ethanol, and is relatively non-toxic, compared to alternative solvents. Thus, it is used widely as a solvent and as a cleaning fluid, especially for dissolving oils.At what temp does isopropyl alcohol evaporate?
This means that pure isopropyl alcohol has a substantial fraction in the gas phase at human physiological temperature (37 deg C). If convection (wind currents) displace the isopropyl alcohol vapor directly above the skin, more of the liquid phase will evaporate to re-establish equilibrium.What temp will alcohol evaporate?
Since alcohol evaporates at 172°F (78°C), any sauce or stew that is simmering or boiling is certainly hot enough to evaporate the alcohol.How fast does alcohol evaporate?
As a reference, here's a helpful rule of thumb: After 30 minutes of cooking, alcohol content decreases by 10 percent with each successive half-hour of cooking, up to 2 hours. That means it takes 30 minutes to boil alcohol down to 35 percent and you can lower that to 25 percent with an hour of cooking.Does rubbing alcohol evaporate water?
Why does water evaporate more slowly than alcohol (and many other liquids)? Ethyl (rubbing) alcohol, with its more loosely bound molecules, evaporates almost five times as quickly as water. When energetic molecules depart from a liquid, they leave lower-energy, lower-temperature molecules behind.How long does it take isopropyl to evaporate?
I heard you that if you put the bottle of 70% isoproyl in the freezer, the water will freeze and you can get 100% isopropyl. I usually wait a few min to be sure but it's usually gone around 30sec.What makes alcohol evaporate faster?
As alcohol evaporates at a much faster rate compared with water due to its lower boiling temperature (82 compared to 100 degrees C), it is able to carry away more heat from the skin. This means for a given amount of time much more alcohol evaporates than water.How long does it take for Everclear to evaporate?
The 95% ethanol will evaporate within a day or two on low heat. The 5% h2o will take the longest.What is the difference between rubbing alcohol and isopropyl alcohol?
Rubbing alcohol is an antiseptic, which contains as not less than 68% and not more than 72% of isopropyl alcohol. The difference between rubbing alcohol and more pure forms of isopropyl alcohol is that rubbing alcohol contains denaturants which make the solution unpalatable for human consumption.How do you make an ice pack without alcohol?
Instructions: Fill the plastic freezer bag with 1 cup of rubbing alcohol and 2 cups of water. Try to get as much air out of the freezer bag before sealing it shut.Homemade gel ice packs
- 1 quart or 1 gallon plastic freezer bags (depending on how large you want the cold pack)
- 2 cups water.
- 1 cup rubbing alcohol (70%)