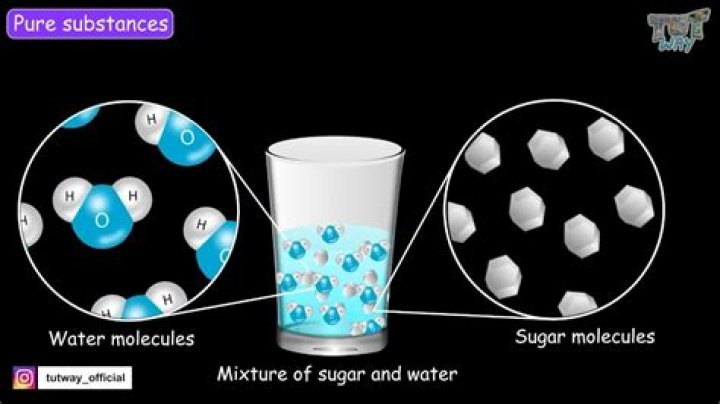
Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. When one of these solids dissolves in water, the ions that form the solid are released into solution, where they become associated with the polar solvent molecules..
In this way, how soluble is sugar in water?
Sugar is pretty soluble in water, but there's a limit. If you add too much sugar to the water, you'll see grains at the bottom no matter how long you stir the mixture. Like sugar and salt, some pesticides are very soluble in water. They dissolve easily.
Also, why is sugar more soluble than salt in water? In a solution, the solute is the substance that dissolves, and the solvent is the substance that does the dissolving. For a given solvent, some solutes have greater solubility than others. For example, sugar is much more soluble in water than is salt. If you add more sugar than this, the extra sugar won't dissolve.
Regarding this, what is it called when sugar dissolves in water?
When you stir a spoonful of sugar into a glass of water, you are forming a solution. This type of liquid solution is composed of a solid solute, which is the sugar, and a liquid solvent, which is the water. As the sugar molecules spread evenly throughout the water, the sugar dissolves.
What causes solubility in water?
The solubility of a given solute in a given solvent typically depends on temperature. For many solids dissolved in liquid water, solubility tends to correspond with increasing temperature. As water molecules heat up, they vibrate more quickly and are better able to interact with and break apart the solute.
Related Question Answers
What is insoluble in water?
insoluble. That's because oil is insoluble or is not capable of being dissolved. Insoluble comes from the Latin insolubilis meaning "that cannot be loosened." When a substance is insoluble, it cannot be dissolved or loosened in water.What is very soluble in water?
Very soluble compounds - definition. Usually materials are treated as sparingly soluble if 1g of material requires 1ml or less of solute to dissolve. In other words, a material will be very soluble if 1ml of solvent will dissolve one or more grams of solute.Why is nacl soluble in water?
Salt (sodium chloride) is made from positive sodium ions bonded to negative chloride ions. Water can dissolve salt because the positive part of water molecules attracts the negative chloride ions and the negative part of water molecules attracts the positive sodium ions.Why sugar can dissolve in water?
Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. When one of these solids dissolves in water, the ions that form the solid are released into solution, where they become associated with the polar solvent molecules.Is caco3 soluble in water?
Calcium carbonate has a very low solubility in pure water (15 mg/L at 25°C), but in rainwater saturated with carbon dioxide, its solubility increases due to the formation of more soluble calcium bicarbonate. Calcium carbonate is unusual in that its solubility increases as the temperature of the water decreases.Which is more soluble in water baking soda or sugar?
Some solutes have greater solubility than others in a given solvent. For example, table sugar is much more soluble in water than is baking soda. You can dissolve much more sugar than baking soda in a given amount of water.Is ethanol soluble in water?
Ethanol has a polar –OH group, which hydrogen bonds to water; which makes ethanol soluble. Ethane which consists of Ions of carbon and hydrogen only has no polar group and is not water-soluble.Why Some salts are insoluble in water?
Insoluble salts are ionic compounds that are insoluble in water: the salt continues to exist as a solid rather than dissolving in the liquid. When a salt such as sodium chloride (table salt) dissolves in water, its ionic lattice is pulled apart so that the individual sodium and chloride ions go into solution.What happens when you dissolve sugar in water?
When sugar dissolves in water, the weak bonds between the individual sucrose molecules are broken, and these C12H22O11 molecules are released into solution. Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules.What is the quickest way to dissolve sugar in water?
If you add a sugar cube to a cup of room-temperature water and another sugar cube to a cup of hot water, you'll find that the sugar dissolves faster in the cup of hot water. The sugar particles move around and interact more at higher temperatures because the additional heat adds more energy to the process.What do you make when you dissolve sugar in water?
A solution is a uniform mixture of a solute (usually a solid) dissolved in a solvent (usually a liquid). When you stir a spoonful of sugar into a glass of water, you are forming a solution.Will sugar dissolve in water?
Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. We can generally assume that salts dissociate into their ions when they dissolve in water.Does sugar dissolve in vodka?
Sugar does not dissolve very well in alcohol because alcohol has a large part that is pretty non-polar. Sugar hardly dissolves at all in oil because oil is very non-polar.Why is water called the universal solvent?
And, water is called the "universal solvent" because it dissolves more substances than any other liquid. This is important to every living thing on earth. This allows the water molecule to become attracted to many other different types of molecules.How do you know when sugar is dissolved in water?
To test if the sugar is completely dissolved: Using spoon, scoop up a small amount of the syrup. You should not be able to see any sugars crystals in the liquid. If you do, boil a little longer.Is wax polar or nonpolar?
Paraffin waxes are insoluble because of its non-polar characteristic. All alkanes are non-polar in nature. And since we always say that "like dissolves like", water being a polar molecule, cannot dissolve paraffin waxes.Is AgCl soluble in water?
Many ionic solids, such as silver chloride (AgCl) do not dissolve in water. The forces holding the solid AgCl lattice together are too strong to be overcome by the forces favoring the formation of the hydrated ions, Ag+(aq) and Cl-(aq).What is more soluble in water salt or sugar?
In other words, a greater weight of sugar will dissolve in a fixed quantity of water than will salt in that same amount of water. Simpler still, sugar is more soluble than salt. Sugar melts easily because the bonds that hold those molecules together in the solid (crystalline state) are much weaker.Which is most soluble in water?
Among given compounds, ethylene glycol ( HO−CH2−CH2−OH ) is the most soluble in water. Ethylene glycol has two hydroxy groups both of which form hydrogen bonds with water. Greater is the number of hydrogen bonds, greater is the extent of hydrogen bonding and greater is the solubility in water.