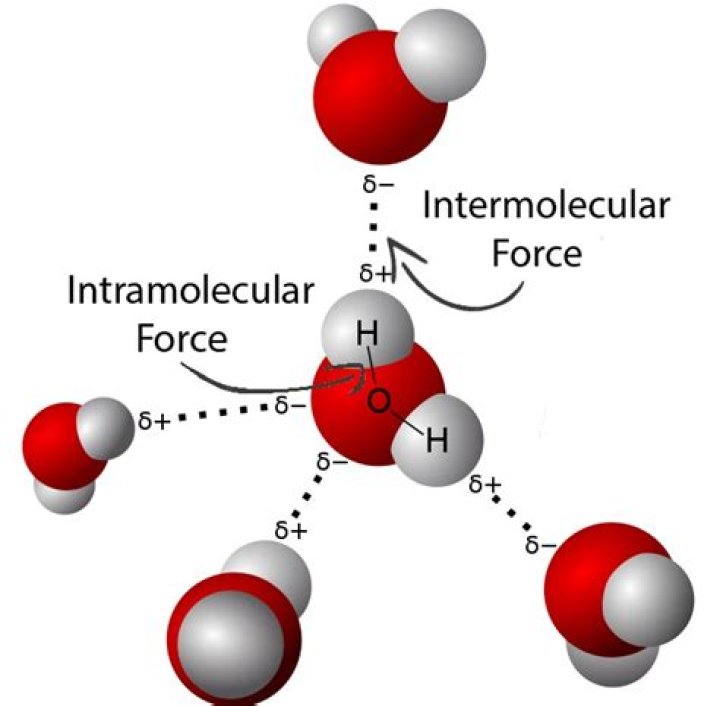
H20 has stronger intermolecular dipole--dipole interaction because of the greater polarity (or a hydrogen bond). Because of the stronger intermolecular interaction H20 is more stable and exists as a liquid. H2S is bent, and bent at a smaller angle than is water..
Also question is, what is the strongest intermolecular force in h2s?
All will exhibit the dispersion force, with H2Te being the strongest with the most electrons. H2S, H2Se and H2Te exhibit dipole-dipole intermolecular forces while H2O exhibits hydrogen bonding. In this case the hydrogen bonding of water is stronger than the dispersion of H2Te.
Likewise, does h2s or h2o have a higher vapor pressure? 14. Water molecules experience hydrogen bonds whereas hydrogen sulfide (H2S) molecules experience dipole forces. Check all of the statements below that are true: a) Water's intermolecular forces are stronger than H2S 's. b) Water has a higher vapor pressure compared to H2S.
Likewise, people ask, which has a greater dipole moment h2o or h2s?
H2S has higher dipole moment than H2O because the bond angle is less in H2S molecule. H2O higher dipole moment than H2S because of the fact that oxygen is more electronegative than sulphur due to which O pulls the electron more than S.
When compared to h2s h2o has a higher boiling point because h2o contains stronger?
Hydrogen bond is more strong than Van Der Waals Force(Present in H2S), so H20 is more stable. During boiling, one has to break the bonds and hence greater amount of energy is required in case of H20 since it is more stable and thus it has a higher boiling point.
Related Question Answers
Is ClF dipole dipole?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). The intermolecular interactions will be made up of dipole-dipole interactions and dispersion interactions.Is h2 a dipole dipole force?
A hydrogen bond is a dipole-dipole force and is an attraction between a slightly positive hydrogen on one molecule and a slightly negative atom on another molecule. Hydrogen bonds are important in the properties of water and in certain biological molecules, such as proteins.Which type of bond is in h2s molecule?
The bonds of H2S are covalent because hydrogen has electronegativity about 2.2, and sulfur 2.56. Because hydrogen has smaller electronegativity it is reducer and sulfur oxidizer. However their difference is smaller, the two have electronegativities for non -metals.Is h2s a dipole?
Since S is more electronegative than H, each S – H bond is polarized with the bond moments directed as shown. Since H2S is a bent molecule the vectorial sum of the bond dipole moments will produce a non- zero total dipole moment. Since the permanent dipole moment is NON-ZERO, H2S will show dipole-dipole interactions .Is co2 a dipole dipole force?
The only intermolecular forces present in CO2 are Van der Waals . All atoms and molecules have Van der Waals forces, so these are present in CO2 . The next strongest are permanent dipole-dipole interactions, which are present between polar molecules.What type of intermolecular force is HCl?
dipole dipole interaction
What are the types of intermolecular forces?
The three major types of intermolecular interactions are dipole–dipole interactions, London dispersion forces (these two are often referred to collectively as van der Waals forces), and hydrogen bonds.What type of intermolecular force is h2o?
Water molecules for example, are held together by hydrogen bonds between the hydrogen atom of one molecule and the oxygen atom of another (fig:hydrogen bonds). Hydrogen bonds are a relatively strong intermolecular force and are stronger than other dipole-dipole forces.Is HF a dipole?
A good example is HF (this is also an example of a special type of dipole-dipole force called a hydrogen bonding). In HF, the bond is a very polar covalent bond. Any molecule with a permanent dipole has dipole-dipole forces that hold the molecules next to each other as a solid or liquid.What is the dipole moment of h2o?
1.8546 d
How do you find the dipole moment of h2o?
The dipole moment is calculated by looking up the dipole moment contributions from each O−H bond, which are polar, and summing them to get the net dipole. Each contribution is 1.5 D (debyes). The net dipole points through oxygen down the y-axis in the negative direction.Why is the dipole moment of ch4 zero?
dipole moment of ch4 is zero . the ch4 is tetrahedral in shape thus each bond pair are at equal distance that is they are symmetrically arranged hence each dipole moment of bond balance each other.Does so2 have a dipole moment?
The SO2 molecule has a dipole moment. This molecule does not have a permanent dipole moment (i.e., the dipole moment of CO2 is zero). The requires the molecule to be linear because oxygen is more electronegative than carbon and therefore the C=O. bond will be polarized.Which molecule has the highest dipole moment?
NaCl
Does SiF4 have a dipole moment?
SiF4 is tetrahedral so that the individual dipoles on the Si-F bonds cancel and the molecule has no dipole moment. S is more electronegative than Te so the partial negative charge on S is considerably bigger leading to a larger dipole moment.What does dipole moment mean?
A dipole moment is a measurement of the separation of two opposite electrical charges. Dipole moments are a vector quantity. The magnitude is equal to the charge multiplied by the distance between the charges and the direction is from negative charge to positive charge: μ = q · r.Is h2s polar or nonpolar?
Hydrogen sulfide is non-polar on account of its non polar H–S bonds. The EN difference between hydrogen and sulfur is 0.4, so hydrogen and sulfur form non-polar bonds. Although it has an asymmetrical molecular geometry, the entire molecule is non-polar dues to the absence of any polar bonds.What determines boiling point?
The boiling point of a liquid is the temperature at which its vapor pressure is equal to the pressure of the gas above it. The normal boiling point of a liquid is the temperature at which its vapor pressure is equal to one atmosphere (760 torr).Is h2o dipole dipole or hydrogen bond?
The H2O water molecule is polar with intermolecular dipole-dipole hydrogen bonds. As the water molecules attract each other and form bonds, water displays properties such as high surface tension and a high heat of vaporization.