Zinc reacts with NaOH to form sodium zincate with the evolution of hydrogen gas..
Consequently, which gas is evolved when sodium hydroxide reacts with zinc?
Hydrogen gas
what are the products obtained when zinc metal is allowed to react with dilute solution of sodium hydroxide? When zinc metal is allowed to react with dilute solution of sodium hydroxide, sodium zincate is formed and hydrogen gas is evolved.
Just so, what happens when metals react with sodium hydroxide?
When alkali (base) reacts with metal, it produces salt and hydrogen gas. Example: Sodium hydroxide gives hydrogen gas and sodium zincate when reacts with zinc metal. Sodium aluminate and hydrogen gas are formed when sodium hydroxide reacts with aluminium metal.
What happens when zinc reacts with potassium hydroxide?
Zinc react with potassium hydroxide and water to produce potassium tetrahydroxozincate(II) and hydrogen.
Related Question Answers
Does NaOH react with zinc?
For exanple, sodium hydroxide reacts with zinc and water to form sodium zincate and hydrogen gas.What happens when Zn reacts with nacl?
Since, zinc is less reactive than sodium, thus no reaction takes place when zinc granules are treated with sodium chloride. Sodium zincate and hydrogen gas are formed when zinc granules are treated with sodium hydroxide and water. On adding dilute hydrochloric acid to the reaction mixture, white precipitate disappears.Why does Zinc react with NaOH?
Zinc is a transition metal which shows moderate reactivity. Zinc reacts with NaOH to form sodium zincate with the evolution of hydrogen gas.How does Zinc react with sodium hydroxide solution?
Zinc react with sodium hydroxide to produce zincate sodium and hydrogen. This reaction takes place at a temperature near 550°C.What happens when zinc granules react with NaOH?
When sodium hydroxide solution is heated with zinc granules then Sodium zincate and hydrogen gas are formed. The salt formed by the reaction between a base and a metal, the metal is present as a part of the Negative Ion . All the metals do not react with bases to form salt and hydrogen gas.Does Zinc react with HCl?
The metal zinc readily reacts with hydrochloric acid to produce hydrogen gas (H2) and zinc chloride (ZnCl2). The reagents are zinc and hydrochloric acid, and the sum is 0 + 2 * ( -167.2) = -334.3.Why does Zinc react with bases?
Accepted Answer: Most metals do not react with bases but zinc metal does because it is amphoteric. That is, it reacts with acids as well as bases. When sodium hydroxide solution is heated with zinc, then sodium zincate and hydrogen gas are formed.Is zinc oxide soluble in sodium hydroxide?
It is nearly insoluble in water, but it will dissolve in most acids, such as hydrochloric acid: ZnO + 2 HCl → ZnCl2 + H2O. Solid zinc oxide will also dissolve in alkalis to give soluble zincates: ZnO + 2 NaOH + H2O → Na2[Zn(OH)4]Which metal does not react with sodium hydroxide?
Aluminium will react vigorously. On the other hand, noble metals like gold or platinum will not react with sodium hydroxide.What metals react with sodium hydroxide?
Sodium hydroxide reacts well both with metals (zinc, aluminum, titanium) and non-metals: halogens, sulfur and phosphorus. In the interaction of sodium hydroxide with aluminum, sodium tetrahydroxoaluminate and hydrogen form. This reaction was used to produce hydrogen to fill air-ships and air balloons.What forms a precipitate with NaOH?
Aluminum hydroxide is a white precipitate, and will dissolve if more sodium hydroxide is added to the solution containing the precipitate. Magnesium hydroxide is also a white precipitate, and will not dissolve if more sodium hydroxide is added to the solution containing the precipitate.What reacts with sodium hydroxide?
Chemical properties The hydroxide ion makes sodium hydroxide a strong base which reacts with acids to form water and the corresponding salts, e.g., with hydrochloric acid, sodium chloride is formed: NaOH( aq) + HCl(aq) → NaCl(aq) + H2O( l)Do all metals react with bases?
All the metals do not react with bases to form salt and hydrogen. Some non-metals react with bases but no hydrogen gas is produced. Some metals are chemically very reactive whereas other metals are less reactive or un reactive.What happens when water reacts with sodium hydroxide?
Reaction of sodium with water Sodium metal reacts rapidly with water to form a colourless solution of sodium hydroxide (NaOH) and hydrogen gas (H2). The resulting solution is basic because of the dissolved hydroxide. The reaction is exothermic.Does sodium hydroxide react with metal?
Can NaOH react with all metals? - Quora. TL;DR No, sodium hydroxide will not react with all metals. Sodium hydroxide is a very strongly basic compound, with a high solubility in water (1110 g/L at 293.15 K) and the ability to pull water from the air surrounding it (meaning it is hygroscopic).What reacts with NaOH to form a white precipitate?
A few drops of dilute sodium hydroxide solution react to form a white precipitate with aluminium ions, calcium ions and magnesium ions. However, if excess sodium hydroxide solution is added: the aluminium hydroxide precipitate dissolves to form a colourless solution. the calcium hydroxide precipitate is unchanged.Is sodium hydroxide corrosive to stainless steel?
Both 304 and 316 stainless steel types are resistant to a wide range of concentration and temperature. Below 80 degC they can be considered resistant to any concentration of sodium hydroxide, up to the limit of solubility.How does Zinc react with dilute NaOH?
Bases also react with certain metals like zinc or aluminum for example to produce hydrogen gas. For exanple, sodium hydroxide reacts with zinc and water to form sodium zincate and hydrogen gas. Zn(s)+2NaOH(aq)+2H2O(l)→Na2Zn(OH)4(aq)+H2(g).What happens when zinc reacts with Sulphuric acid?
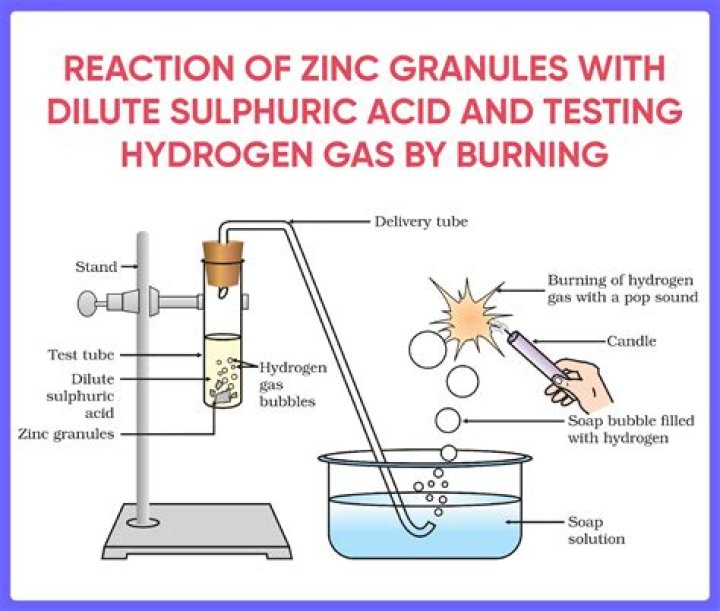
Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved. This is a single displacement reaction of a non-metal by a metal. The zinc will react with sulphuric acid forming zinc sulphate dissolved in solution and releasing hydrogen gas.