Which electron orbital is full for neon?
By Jessica Cortez •
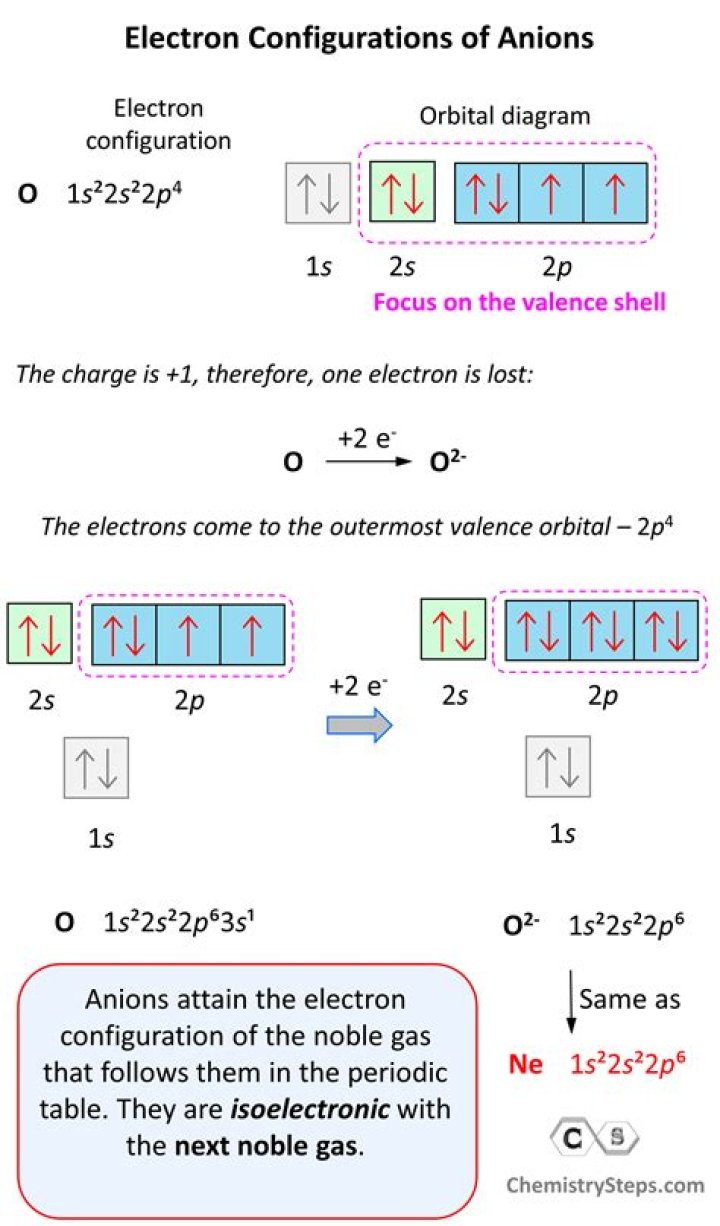
Neon is the tenth element with a total of 10 electrons. In writing the electron configuration for neon the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Ne go in the 2s orbital. The remaining six electrons will go in the 2p orbital.
.
Herein, what is the electron arrangement for neon?
[He] 2s2 2p6
Secondly, how many electrons does neon have? 2,8
Similarly, it is asked, how many orbitals are in neon?
Neon ( Nestart text, N, e, end text), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1s orbital and eight fill the second shell—two each in the 2 s 2s 2s and three p orbitals, 1 s 2 1s^ 2 1s21, s, squared 2 s 2 2s^ 2 2s22, s, squared 2 p 6 2p^6 2p62, p, start superscript, 6, end
What is the electron configuration for neon atomic number 10?
Neon symbol is Ne, atomic number 10 that locate in 2 period of noble gas group. Ne has 20.1797 atomic mass, 10 electrons and protons, 10.1797 neutrons, electron configuration is [He]2S22p6.
Related Question AnswersDoes hydrogen have more electrons than uranium?
Does hydrogen have more electrons than Uranium? yes 9.Why does the D block span 10 groups?
The d-block contains the transition metals and is the largest block. There are exceptions, but d-block elements usually have filled outermost s orbital, and filled or partially filled d orbital. The five d orbitals can hold 10 electrons, so the d-block spans ten groups on the periodic table.What is valence configuration?
The electrons that occupy the outermost shell of an atom are called valence electrons. Valence electrons are important because they determine how an atom will react. By writing an electron configuration, you'll be able to see how many electrons occupy the highest energy level.How many 3d electrons are in FE?
6 electronsIs Neon a metal nonmetal or metalloid?
Neon is neither a metal nor a metalloid. It is a non-metal. Neon is a chemical element that has a symbol of “Ne” and an atomic number 10. It is in fact a noble gas element.Why is neon an unreactive element?
Neon has eight electrons in its outer shell, it doesn't want to donate any, it doesn't want to accept any therefore it is unreactive. All the noble gases have full outer shells, so they are all unreactive. Sometimes the noble gases are called "inert" because of their unreactiveness.Is ground state neutral?
The ground state is the most stable energy state for the electrons of a given atom. A neutral chlorine atom has 17 protons (atomic number 17) and 17 electrons. ( px,py,pz ). The last 7 electrons, the outermost electrons with the highest energy levels, are located in the third energy level.Why is neon in the same group of the periodic table as helium?
Since helium and neon are both in group 18, they are atomically similar. Because noble gases' outer shells are full, they are extremely stable, tending not to form chemical bonds and having a small tendency to gain or lose electrons. Other physical/atomic and chemical properties will also be similar.What is the orbital diagram of neon?
Neon is the tenth element with a total of 10 electrons. In writing the electron configuration for neon the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Ne go in the 2s orbital. The remaining six electrons will go in the 2p orbital.What is the Bohr diagram for neon?
Two electron shells surrounding the nucleus, containing 2 electrons in the n=1 shell and 8 electrons in the n=2 shell. Bohr's model of the atom described the atom as a series of energy levels called principle quantum shells, at progressively greater distance from the nucleus.Which orbital is the first to be filled in any atom?
1sWho discovered orbitals?
However, the idea that electrons might revolve around a compact nucleus with definite angular momentum was convincingly argued at least 19 years earlier by Niels Bohr, and the Japanese physicist Hantaro Nagaoka published an orbit-based hypothesis for electronic behavior as early as 1904.Why are there only two electrons in the first shell?
This first shell has only one subshell (labeled 1s) and can hold a maximum of 2 electrons. This is why there are two elements in the first row of the periodic table (H & He). Because the first shell can only hold a maximum of 2 electrons, the third electron must go into the second shell.Does neon react with anything?
Neon (Ne) is the second of the noble gases. Just like all noble gases, it is very non-reactive. So much so, that it doesn't form compounds with anything. Just like helium (He) and argon (Ar), neon floats around all by itself.What is Hund rule?
Hund's Rule. Hund's rule: every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals have the same spin.How many electrons does neon gain or lose?
These have the same electron configuration as the noble gas neon. Elements in Group 14 could lose four, or gain four electrons to achieve a noble gas structure.What is the number of valence electrons in phosphorus?
fiveHow much does neon cost for 1 gram?
| Name | Neon |
|---|---|
| Normal Phase | Gas |
| Family | Noble Gas |
| Period | 2 |
| Cost | $33 per 100 grams |

