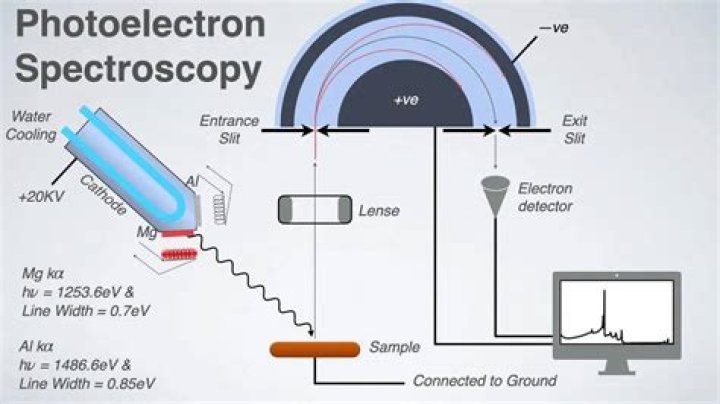
Which agency holds the legal responsibility for deciding whether a drug may be distributed and sold
The Food, Drug, and Cosmetic Act (FDCA) governs interstate distribution of medical products. Manufacturers may distribute drugs and certain devices for commercial use only after the FDA has approved the products as safe and effective for their intended use.
Who is legally responsible for deciding whether a drug may be distributed and sold?
The Food and Drug Administration (FDA), a regulatory agency within the Department of Health and Human Services, regulates the safety and effectiveness of drugs sold in the United States.
What is it called when drugs are injected through a syringe into a muscle vein or body cavity?
With chemo infusions, chemotherapy drugs are put into your body through a thin tube called a catheter that’s placed in a vein, artery, body cavity, or body part. In some cases, a chemo drug may be injected quickly with a syringe.
Which governing agency approves the investigational use of drugs on humans?
The U.S. Food and Drug Administration (FDA) is the federal agency responsible for labeling medications and supplements. However, the approval process is different for prescription and for over-the-counter medications.What is the drug administration route with insertion of suppositories?
Rectal route Many drugs that are administered orally can also be administered rectally as a suppository. In this form, a drug is mixed with a waxy substance that dissolves or liquefies after it is inserted into the rectum.
Who regulates drugs in Canada?
Health Canada’s HPFB is the national authority that regulates, evaluates and monitors the safety, efficacy, and quality of therapeutic and diagnostic products available to Canadians.
Who decides FDA approval?
FDA approval of a drug means that data on the drug’s effects have been reviewed by CDER, and the drug is determined to provide benefits that outweigh its known and potential risks for the intended population.
Why are drugs regulated?
Most governments around the world impose regulations on pharmaceutical companies, in an effort to protect their public from harmful drug effects. These regulations often prolong the process for bringing new pharmaceuticals to market.Who approves clinical trials in India?
The Drugs Controller General of India (DCGI) is an official of the CDSCO who is the final regulatory authority for the approval of clinical trials in the country.
What is IND and NDA?The Investigational New Drug (IND) application falls into the first category, while the New Drug Application (NDA), Abbreviated New Drug Application (ANDA), and Biologics License Application (BLA) fall into the second category.
Article first time published onWhich type of medication is injected with syringe?
An injection is a way of administering a liquid to a person using a needle and syringe. It’s also sometimes also called a ‘shot’ or ‘jab’. Injections are used to give a wide variety of different medications, such as insulin, vaccines and Botox (onabotulinumtoxinA), but not all injections are the same.
Which of the following is the action of a diuretic?
Diuretics, sometimes called water pills, help rid your body of salt (sodium) and water. Most of these medicines help your kidneys release more sodium into your urine. The sodium helps remove water from your blood, decreasing the amount of fluid flowing through your veins and arteries. This reduces blood pressure.
Where is intravenous injection given?
Some medications must be given by an intravenous (IV) injection or infusion. This means they’re sent directly into your vein using a needle or tube. In fact, the term “intravenous” means “into the vein.” With IV administration, a thin plastic tube called an IV catheter is inserted into your vein.
What are drug administration routes?
A route of administration in pharmacology and toxicology is the path by which a drug, fluid, poison, or other substance is taken into the body. Routes of administration are generally classified by the location at which the substance is applied. Common examples include oral and intravenous administration.
How do you administer a suppository?
You can stand with one leg up on a chair or lie on your side with one leg straight and the other bent in toward your stomach. Gently spread your buttocks open. Carefully push the suppository, tapered end first, about 1 inch into your bottom. Close your legs and sit or lie still for about 15 minutes to let it dissolve.
Which of the following routes of administration would be considered parenteral?
Parenteral routes of administration include the subcutaneous, intramuscular, and intravenous routes.
What does NIH do for the FDA?
NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases.
What is required for FDA approval?
To get FDA approval, drug manufacturers must conduct lab, animal, and human clinical testing and submit their data to FDA. FDA will then review the data and may approve the drug if the agency determines that the benefits of the drug outweigh the risks for the intended use.
What does FDA approve?
The FDA regulates a wide range of products, including foods (except for aspects of some meat, poultry and egg products, which are regulated by the U.S. Department of Agriculture); human and veterinary drugs; vaccines and other biological products; medical devices intended for human use; radiation-emitting electronic …
Is Dayvigo approved in Canada?
Health Canada approved the following indication: Dayvigo (lemborexant) is indicated for the treatment of insomnia, characterized by difficulties with sleep onset and/or sleep maintenance.
Does the FDA approve drugs in Canada?
Most health products, including drugs to be marketed or sold in Canada are reviewed and authorized by the Health Products and Food Branch (HPFB) of Health Canada, more precisely, under the Therapeutic Product Directorate (TPD) or the Biologic and Genetic Therapies Directorate (BGTD), for drugs and biologic, …
What does Health Canada regulate?
Health Canada helps make sure companies and products meet Canada’s high safety and quality standards by promoting and enforcing compliance with laws and regulations. We also: carry out targeted or planned reviews of specific product types.
Who is responsible for approving the protocol for a clinical trial?
Before clinical research ensues, it must first be approved by an institutional review board (IRB). When conducting clinical trials, researchers should maintain communication with their study participants.
Who approves clinical trial protocol?
Each federally supported or conducted clinical study and each study of a drug, biological product, or medical device regulated by FDA must be reviewed, approved, and monitored by an institutional review board (IRB). An IRB is made up of doctors, researchers, and members of the community.
What is protocol amendment?
“Protocol Amendment: Change in Protocol” A sponsor of an IND application is expected to submit a protocol amendment in cases when there are changes in the existing protocol that significantly affect safety of subjects, scope of the investigation, or scientific quality of the study.
What are drugs regulatory agencies?
Regulatory authority and organizations are responsible in effective drug regulation required to ensure the safety, efficacy and quality of drugs, as well as the accuracy and appropriateness of the drug information available to the public.
What is the government's responsibility in ensuring that drugs are available to those who need them?
The Food and Drug Administration (FDA) is the U.S. government agency charged with ensuring the safety and efficacy of the medicines available to Americans. … The two legislative acts that are the main source of the FDA’s powers both followed significant tragedies.
Under which rule the roles and responsibilities of drug inspector has been defined?
21. Inspectors. (4) Every Inspector shall be deemed to be a public servant within the meaning of section 21 of the Indian Penal Code, and shall be officially subordinate to such authority [(Note: Ins. …
Who governs pharmacovigilance?
The World Health Organization defines pharmacovigilance (PV) as “the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other drug-related problem.”
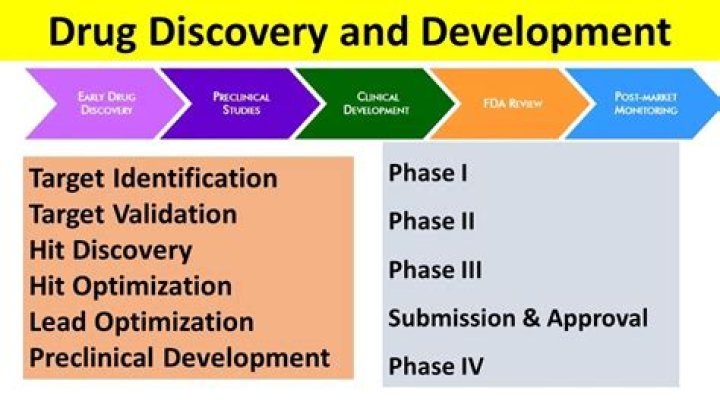
What is IND in drug discovery?
An Investigational New Drug Application (IND) is a request from a clinical study sponsor to obtain authorization from the Food and Drug Administration (FDA) to administer an investigational drug or biological product to humans.
When the sponsor investigator holds the IND for an investigational drug he or she is responsible for?
The Sponsor of the IND or IDE application is responsible for notifying the FDA PROGRAM MANAGER and the IRB immediately of a “clinical hold” issued by the FDA for a clinical investigation being conducted under a University-based IND and IDE application and/or of any other FDA actions or determinations (e.g., FDA ‘483’ …