What reacts with acid to form bubbles?
What reacts with acid to form bubbles?
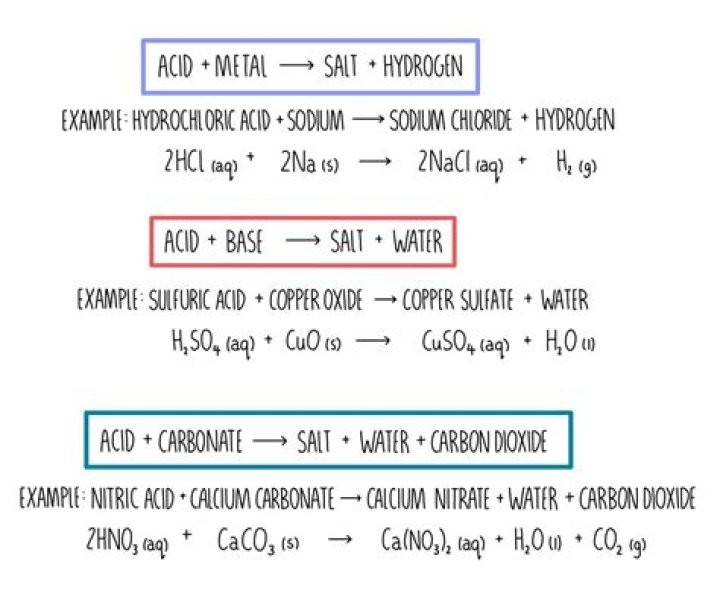
When acids react with carbonates, such as calcium carbonate (found in chalk, limestone and marble), a salt, water and carbon dioxide are made. The carbon dioxide causes bubbling during the reaction, which is observed as fizzing.
What substances can acids react with to produce hydrogen gas?
Dilute acids react with relatively reactive metals such as magnesium, aluminium, zinc and iron. The products of the reaction are a salt plus hydrogen gas.
Does copper react with acid?
Copper is present below hydrogen in the reactivity series of metals. So it does not react with dilute acids like hydrochloric acid or sulfuric acid.
Which of the metals reacts with acids?
Reacting metals and acids Common metals for reacting with acids (in order of reactivity) would be magnesium, zinc, iron, tin and copper; i.e. one from the top, three from the middle and one from the bottom of the reactivity series.
What happens if you add salt to acid?
acid + hydrogencarbonate → salt + water + carbon dioxide The carbon dioxide gas produced in these reactions can be tested.
Which are weak acids?
Now let’s discuss some weak acid examples:
- Acetic acid (CH3COOH)
- Formic acid (HCOOH)
- Oxalic acid (C2H2O4)
- Hydrofluoric acid (HF)
- Nitrous acid (HNO2)
- Sulfurous acid (H2SO3)
- Phosphoric acid (H3PO4)
- Benzoic acid (C6H5COOH)
Is HCl a weak acid?
HCl is a strong acid because it dissociates almost completely. By contrast, a weak acid like acetic acid (CH3COOH) does not dissociate well in water – many H+ ions remain bound-up within the molecule. In summary: the stronger the acid the more free H+ ions are released into solution.
What is the formula of hydrochloric acid?
HCl
Hydrochloric acid/FormulaWhat happen when copper reacts with acid?
Reaction of copper with acids Copper metal dissolves in hot concentrated sulphuric acid to form solutions containing the aquated Cu(II) ion together with hydrogen gas, H2. In practice, the Cu(II) is present as the complex ion [Cu(OH2)6]2+. Copper metal also dissolves in dilute or concentrated nitric acid, HNO3.
Why can’t copper react with acid?
Yes, it is obvious that copper will not react with hydrochloric acid for the simple reason it’s below hydrogen in the reactivity series. So, copper cannot replace the hydrogen in HCl to form CuCl2 . Hence, when copper (Cu) reacts with hydrochloric acid (HCl) there will be no reaction.
What reacts with hydrochloric acid?
These metals — beryllium, magnesium, calcium and strontium — react with hydrochloric acid to form a chloride and free hydrogen. Metallic magnesium when combined with hydrochloric acid, will naturally result in magnesium chloride — used as a dietary supplement — with the hydrogen being released as a gas.
What is Zn HCl?
Zn + HCl → ZnCl2 + H2. Word equation: Zinc chloride + Hydrogen gas → Zinc + Hydrochloric acid. Type of Chemical Reaction: For this reaction we have a combination reaction. Balancing Strategies: When we add zinc to hydrochloric acid we end up with zinc chloride, a salt, and hydrogen gas.
What are the ingredients in a bubble solution?
The three key elements of most bubble solutions are the surfactant (usually provided by dishwashing liquid or some type of soap product), a polymer of some sort, and water. There are often additional ingredients. To understand the role that these ingredients play, please see Bubble Juice Basics . Using the right detergent or soap is critical.
What kind of gas is produced in an acid base reaction?
Both of these reactions can produce a gas, either carbon dioxide or hydrogen sulfide. In the lab, sodium bicarbonate is usually used to neutralize acid spills. When it reacts with acid it produces bubbles, so it’s easy to see when the reaction finishes. Most chemists agree that acid-base reactions are combination reactions without redox.
Why do acid base reactions make cakes fluffy?
This is called neutralization. Acid-base neutralization reactions are what make most cakes fluffy, because sometimes these reactions generate a gas that makes holes in the cake. It is important to know that there are many different definitions of acid and base. This page describes the simplest and most specific definition.
What can you substitute for baking soda in soap bubble?
Sodium citrate (sometimes sold as sour salt) is a byproduct along with CO2 of the reaction between baking soda, citric acid and water. To substitute sodium citrate for baking soda/citric acid, use 1.17 times as much Sodium Citrate as you would baking soda (and eliminate the citric acid). Flax seeds (lin seeds).
The three key elements of most bubble solutions are the surfactant (usually provided by dishwashing liquid or some type of soap product), a polymer of some sort, and water. There are often additional ingredients. To understand the role that these ingredients play, please see Bubble Juice Basics . Using the right detergent or soap is critical.
Both of these reactions can produce a gas, either carbon dioxide or hydrogen sulfide. In the lab, sodium bicarbonate is usually used to neutralize acid spills. When it reacts with acid it produces bubbles, so it’s easy to see when the reaction finishes. Most chemists agree that acid-base reactions are combination reactions without redox.
This is called neutralization. Acid-base neutralization reactions are what make most cakes fluffy, because sometimes these reactions generate a gas that makes holes in the cake. It is important to know that there are many different definitions of acid and base. This page describes the simplest and most specific definition.
What does the reaction between acid and base look like?
The general reaction looks like this: Overall, the reaction is: Thus, the hydrogen ions, which makes acids acidic, are consumed, and the hydroxide which makes bases basic is also consumed, and if the moles of acid and base are equal, only neutral water and a salt is left.

