What is the Zeff of sodium?
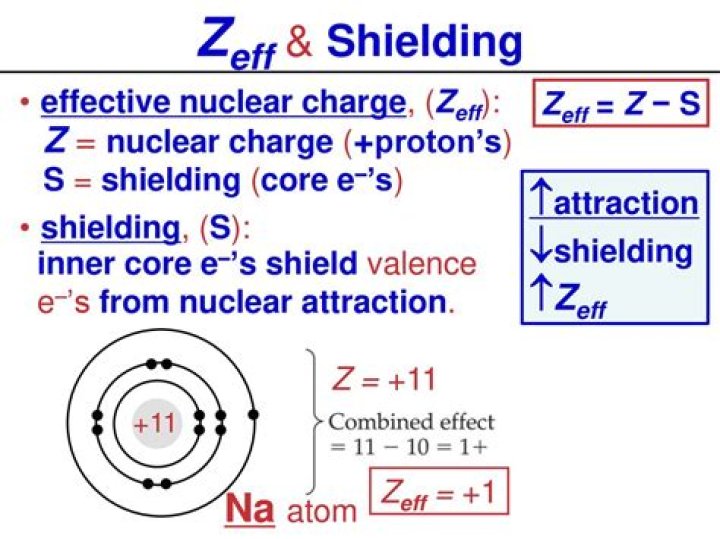
What is the Zeff of sodium?
Step 5: Find Z Effective Using Formula The effective nuclear charge of the 3s1 electron in the sodium atom is 2.2. Note the value is a charge and contains no units.
How do you calculate z effective?
Subtract S from Z Finally subtract the value of S from Z to find the value of effective nuclear charge, Zeff. For example, Us the Lithium atom, then Z =3 (atomic number) and S = 1.7. Now put the variables in the formula to know the value of Zeff (effective nuclear charge).
What is the Z effective of potassium?
Calculate the effective nuclear charge experienced by the 4s-electron in potassium atom (Z=19).
What is the Z effective of fluorine?
Fluorine: Zeff = 9 – 2 = 7+. So: when we go down a period the effective nuclear charge increases. Example: atomic radius of C vs F. Zeff of C is 4+, and C has 4 valence electrons.
How do you calculate Sigma in Zeff?
Formula for S & P – electrons: Sigma = (n-1)*0.35 + (n’)*0.85 + (n”)*1. (2-1)*0.35 + 14*0.85 + 10*1 = 23.25. Zeff = 26-23.25 = 2.75.
Why does Z effective increase down a group?
A higher effective nuclear charge causes greater attractions to the electrons, pulling the electron cloud closer to the nucleus which results in a smaller atomic radius. Down a group, the number of energy levels (n) increases, so there is a greater distance between the nucleus and the outermost orbital.
What is the Z effective of scandium?
Here, is the ‘effective nuclear’ charge and Z is the atomic number and σ is the shielding factor. Thus, the effective nuclear charge for 4s and 3d electrons in Sc is 1.65 and 3 respectively.
What is the Z effective of rubidium?
Effective nuclear charge of Rubidium: Atomic number of rubidium is 37. According to slater rules: electrons in the same shell contribute 0.35, electrons in (n-1) shell contribute 0.85 and electrons in (n-2) or lower shells contribute 1.00 to the total shielding constant.
What is the effective nuclear charge Z * for the single H electron?
Truong-Son N. and the effective nuclear charge is Zeff=Z=1 , where Z is the atomic number.

