What is the structure of a Krypton-85 atom?
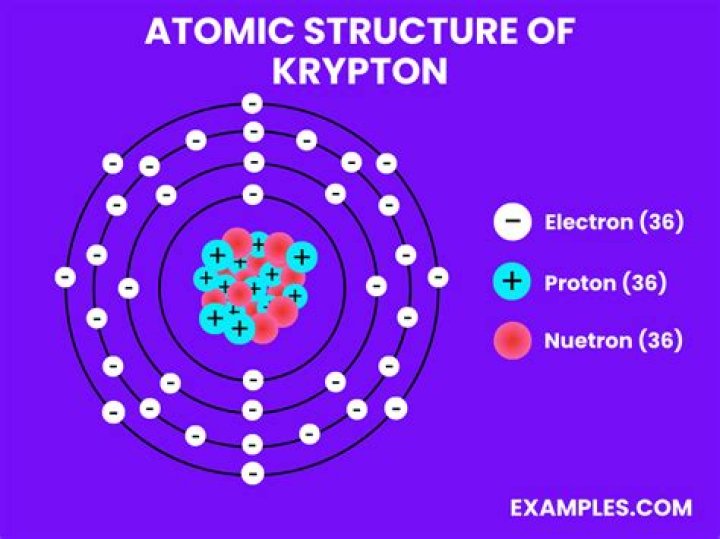
What is the structure of a Krypton-85 atom?
Krypton-85 (85Kr) is a radioisotope of krypton….Krypton-85.
| General | |
|---|---|
| Names | krypton-85, Kr-85 |
| Protons | 36 |
| Neutrons | 49 |
| Nuclide data |
What is the difference between Krypton and Krypton-85?
Krypton-81 has a half-life of 210,000 years, and krypton-85 has a half-life of 11 years; the half-lives of the other krypton isotopes are less than two days. Krypton-85 is the isotope of concern at Department of Energy (DOE) environmental management sites such as Hanford.
How is Krypton-85 formed?
Atmospheric Krypton-85 is mostly produced by volcanoes, earthquakes, nuclear power plants and nuclear explosions. Since mid-1940s, much greater quantities of Krypton-85 than that found in nature have been manufactured as a byproduct of nuclear fission.
What does the number 85 in the name Krypton-85 represent?
The Krypton-85 nucleus (the 85 means there are a total of 85 protons and neutrons in the atom) spontaneously turns into a nucleus of the element Rubidium which still has a sum of 85 protons and neutrons, and a beta particle (electron) flies out, resulting in no net difference in charge.
What is krypton-85 Halflife?
properties of krypton …contains only one radioactive isotope, krypton-85, which has a half-life of 10.7 years, because all the other radioactive isotopes have half-lives of 3 hours or less.
Is krypton-85 a gas?
Description Krypton-85 is a radioactive gas found in the atmosphere and produced by nucliear explosions, nuclear power plants, volcanoes and earthquakes. Krypton-85 is odorless, colorless and tasteless and emits low level radiation levels of both gamma and beta rays.
Is krypton 85 a gas?
Is Krypton 81 stable or unstable?
Isotopes of krypton
| Isotope | ||
|---|---|---|
| abundance | half-life (t1/2) | |
| 80Kr | 2.29% | stable |
| 81Kr | trace | 2.3×105 y |
| 82Kr | 11.59% | stable |
Who discovered krypton 85?
Sir William Ramsay
Krypton was discovered on May 30, 1898 by Sir William Ramsay, a Scottish chemist, and Morris M. Travers, an English chemist, while studying liquefied air. Small amounts of liquid krypton remained behind after the more volatile components of liquid air had boiled away.
What are krypton’s uses?
Krypton is used commercially as a filling gas for energy-saving fluorescent lights. It is also used in some flash lamps used for high-speed photography. Unlike the lighter gases in its group, it is reactive enough to form some chemical compounds. For example, krypton will react with fluorine to form krypton fluoride.
How many electrons does krypton 85 have?
What is the structure of a krypton 85 atom A 49 electrons 49 protons 85 neutrons | Course Hero.
How is the radioactive decay of krypton 85?
The stable value is closer to 1.3, so Kr−85 decays by β− emission. The atomic number increases by 1 unit, but the mass number stays the same. There are now 37 protons and 48 neutrons. The product is the stable, non-radioactive nuclide, rubidium-85.

