What is the role of base in Suzuki reaction?
What is the role of base in Suzuki reaction?
Hence, the main role of the base in the reaction mechanism is to increase the reactivity of the boronic acid toward the Pd–halide complex by converting it into the respective organoborate.
Which step involves in Suzuki coupling reaction?
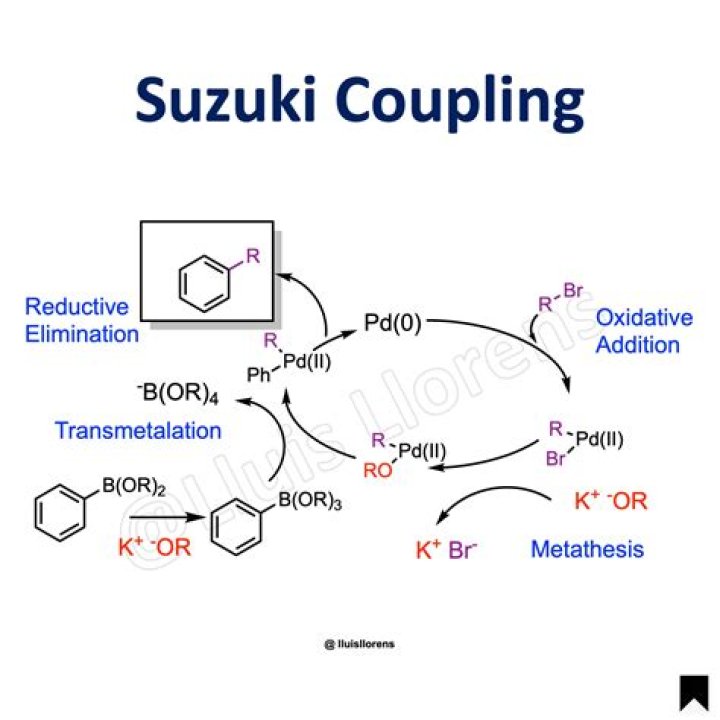
Suzuki Coupling Reaction Mechanism The Suzuki coupling mechanism follows a catalytic cycle involving three primary steps, namely – oxidative addition, transmetalation, and reductive elimination.
Why is palladium used in coupling reactions?
Generally, the reactivity of organopalladium complexes is lower compared with organonickel complexes. However, they have higher chemical stability for oxidations and this makes them easy to use. Therefore, palladium complexes are most commonly used for cross-coupling reactions.
What is the purpose of hydroxide ion in the Suzuki reaction?
Three roles for hydroxide: The base OH− plays a triple role in Suzuki–Miyaura reactions (see graphic): 1) formation of [ArPd(OH)(PPh3)2] as the reactive species involved in the rate-determining transmetalation, 2) formation of unreactive Ar′B(OH)3− by reaction of OH− with ArB(OH)2, and 3) unexpected acceleration of the …
Which base is used in Suzuki coupling?
The general scheme for the Suzuki reaction is shown below, where a carbon-carbon single bond is formed by coupling an organoboron species (R1-BY2) with a halide (R2-X) using a palladium catalyst and a base….
| Suzuki reaction | |
|---|---|
| Organic Chemistry Portal | suzuki-coupling |
| RSC ontology ID | RXNO:0000140 |
Which is suitable catalyst for the Suzuki coupling reaction?
Palladium catalysts are the most widely used for Suzuki coupling and perform best with electron-donating (usually phosphine) ligands. Nickel catalysts have been recently developed and demonstrate reactivity with inert electrophiles, especially chlorides and unreactive bromides.
What is Suzuki coupling used for?
This reaction is also known as the Suzuki–Miyaura reaction or simply as the Suzuki coupling. It is widely used to synthesize polyolefins, styrenes, and substituted biphenyls. Several reviews have been published describing advancements and the development of the Suzuki reaction.
Why is Suzuki coupling important?
It is also known as the Suzuki–Miyaura reaction or Suzuki coupling. The reaction is important to organic chemistry because it forms carbon-carbon bonds allowing the synthesization of various organic molecules. The discovery of palladium-catalyzed cross couplings was awarded the 2010 Nobel Prize in Chemistry.
What is palladium catalyst?
palladium (Pd), chemical element, the least dense and lowest-melting of the platinum metals of Groups 8–10 (VIIIb), Periods 5 and 6, of the periodic table, used especially as a catalyst (a substance that speeds up chemical reactions without changing their products) and in alloys.
How does palladium work as a catalyst?
Carbon atoms meet on a palladium atom, and then are so close together that chemical reactions are possible. In “palladium-catalyzed cross coupling,” palladium acts as a catalyst. It “encourages” and “enables” a reaction between individual carbon atoms.
What is the role of water in Suzuki coupling?
These water molecules form as a by-product of a side reaction, the trimerization of phenylboronic acid. This small amount of water can play a key role in the desired transformation, making it a pseudo-solid-state reaction. The team reacted solid aryl halides with phenylboronic acid in the absence of any liquids.
How Organohalides are combines with palladium catalyst in cross-coupling reaction?
Oxidative addition between the organohalide and Pd(0) catalyst results in the formation of an organopalladium(II) species. This organopalladium(II) species reacts with the nucleophilic organometallic compound in a step called transmetalation to generate an organopalladium(II) species with two carbon-palladium bonds.