What is the purpose of redox reactions?
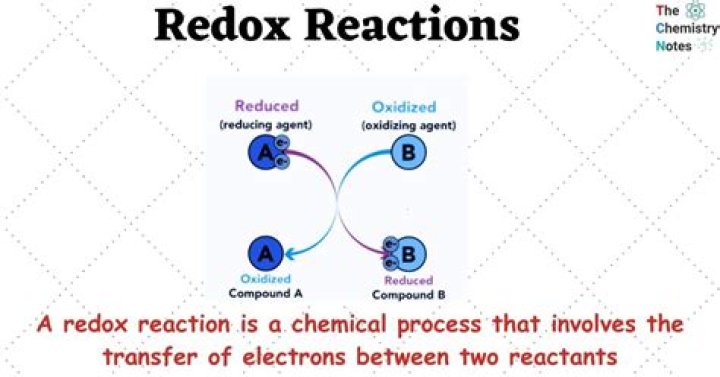
What is the purpose of redox reactions?
Oxidation-reduction (redox) reactions are important because they are the principal sources of energy on this planet, both natural or biological and artificial. Oxidation of molecules by removal of hydrogen or combination with oxygen normally liberates large quantities of energy.
What is a spontaneous redox reaction?
A spontaneous redox reaction is a type of reaction in which there is usually the release of energy where the electrons are transferred from let’s say anode to cathode. This type of reaction is mostly seen in electrochemical cells. Here, the reaction leads to the generation of electricity.
Why is a redox reaction spontaneous?
A redox reaction is spontaneous if the standard electrode potential for the redox reaction, Eo(redox reaction), is positive. If Eo(redox reaction) is positive, the reaction will proceed in the forward direction (spontaneous). And non-spontaneous redox reactions cannot be used to produce electricity.
Where are redox reactions used?
A few examples of redox reactions in everyday life are discussed in terms of photosynthesis, decay, respiration, biological processes, corrosion/rusting, combustion and batteries. produced as fuel for its metabolic process. The reaction of photosynthesis is sensitized by chlorophyll.
What is the role of redox reaction to human lives?
Respiration Cellular respiration which is the ultimate source of energy in human beings encompasses a series of redox reactions. So, the food that we consume is converted into energy by redox reactions only. During the process of respiration, the carbon-dioxide is reduced whereas the water is oxidised to form oxygen.
What are combination reactions?
A combination reaction is a reaction in which two reactants combine to form one product. Oxygen and the halogens are very reactive elements and are likely to undergo combination reactions with other elements.
What is spontaneous and non spontaneous redox reaction?
A spontaneous redox reaction generates a voltage itself. A nonspontaneous redox reaction occurs when an external voltage is applied. The reactions that occur in an electric battery are electrochemical reactions.
What is spontaneous reaction and non spontaneous reaction?
A spontaneous process is capable of proceeding in a given direction without needing to be driven by an outside source of energy. An endergonic reaction (also called a nonspontaneous reaction) is a chemical reaction in which the standard change in free energy is positive and energy is absorbed.
How do we know if a reaction is spontaneous?
If ΔH is negative, and –TΔS positive, the reaction will be spontaneous at low temperatures (decreasing the magnitude of the entropy term). If ΔH is positive, and –TΔS negative, the reaction will be spontaneous at high temperatures (increasing the magnitude of the entropy term).
How are redox reactions used in industry?
Oxidation processis used in the industrial production of cleaning products. Nitric acid which is a component in many fertilizers is produced from the oxidation reaction of ammonia. Redox reactions are also used in in the process of electroplating by applying a thin coating of a material on an object.
What are the uses of combination reaction?
Combination reactions can be used to prepare a variety of stable ionic compounds. If you know what the most stable ions of various elements are you can predict the formulae for these ionic compounds.
Why combination reactions are known as exothermic reactions?
Exothermic reactions are the reaction in which energy is released in the form of heat…in combination reaction energy is released during the formation of bond… hence it is exothermic..

