silver
.
Keeping this in view, what is the most conductive metal?
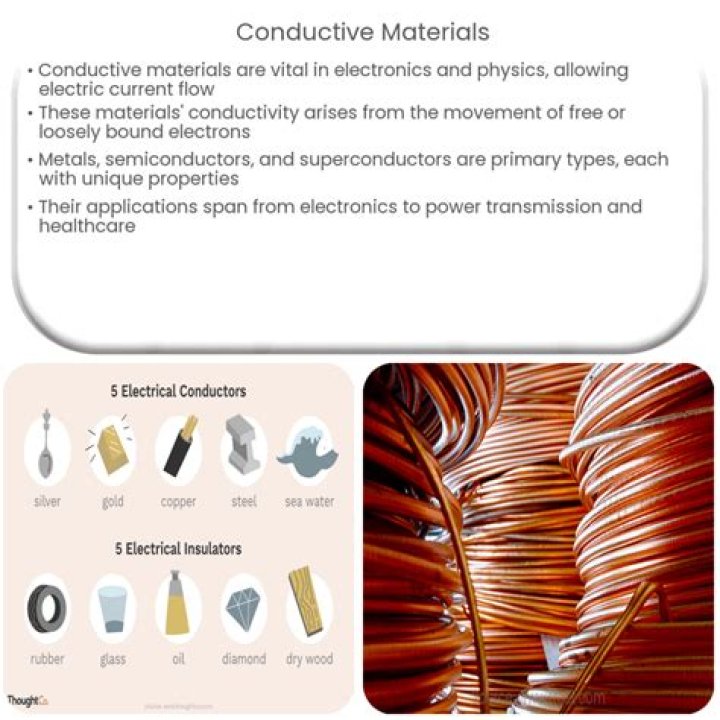
The most highly conductive metals are silver, copper, and gold. Copper, for example, is highly conductive and commonly used in metal wiring. Brass, on the other hand, contains copper but other materials in its makeup reduce conductivity. Pure silver is the most conductive of all metals.
Furthermore, what metal is the least conductive? Stainless steel
Additionally, which metal is best electrical conductor?
Silver
Is metal a good conductor of electricity?
Metals are good conductors of electricity since the metallic bond gives rise to the” free electrons “ where outer orbit electrons of metal atoms are shared by many adjacent atoms and they are “loosely bound.
Related Question Answers
Which metal is the poorest conductor of electricity?
Bismuth and tungsten are two metals which are poor conductors of electricity. There are many, but some include Aluminum, Bismuth, Gallium, Indium, Lead, Thallium, Tin, Ununhexium, Ununpentium, Ununquadium, and Ununtrium.What makes a metal more conductive?
Electrical conductivity in metals is a result of the movement of electrically charged particles. It is these "free electrons" that allow metals to conduct an electric current. Because valence electrons are free to move, they can travel through the lattice that forms the physical structure of a metal.Is stainless steel electrically conductive?
Stainless steel is a relatively good conductor of electricity, as are all metals. The body is also a good conductor, due to water. The skin is a relatively poor conductor of electricity because of dead skin cells. Any break in the skin, i.e. cut, greatly reduces its resistance.Is graphite a good conductor of electricity?
Graphite is an interesting material, an allotrope of carbon (as is diamond). It displays properties of both metals, and nonmetals. However, like a metal, graphite is a very good conductor of electricity due to the mobility of the electrons in its outer valence shells.Which metal can be cut with knife?
Sodium
Is copper more conductive than stainless steel?
Copper is much more thermally conductive than aluminum, admiralty brass, carbon steel, and stainless steel. Stainless steel's low thermal conductivity is partially counteracted with the ability to have thinner walls because of its high strength and corrosion resistance.Is aluminum a precious metal?
An initially precious metal that became common is aluminium. While aluminium is the third most abundant element and most abundant metal in the Earth's crust, it was at first found to be exceedingly difficult to extract the metal from its various non-metallic ores. By that time, aluminium was as expensive as silver.Is glass a conductor?
Glass is actually an insulator. It doesn't allow the flow of electrons easily from atom to atom, as seen in substances like copper, and other metals which are excellent conductors of both heat and electricity. Glass, wood and plastic are all excellent insulators, but not good conductors.Who is the good conductor of electricity?
Silver also has the highest thermal conductivity of any element and the highest light reflectance. Although it is the best conductor, copper and gold are used more often in electrical applications because copper is less expensive and gold has a much higher corrosion resistance.Is Diamond a good conductor of electricity?
Diamond is a good conductor of heat but bad conductor of electricity. Electric current is caused due to availablity of free electrons in a crystal . In case of diamond, each carbon atom is covalently bonded with four other carbon atoms and hence no free electrons are available to conduct electric current.Which metal is a bad conductor of electricity?
Tungsten and Bismuth are metals which are poor conductors of electricity. Stainless Steel is a poor conductor because it has an alloy structure.Is gold a good conductor of electricity?
Gold is a very good conductor of electricity. Even though copper and silver are better conductors than gold, gold retains its conductivity longest because it does not tarnish or corrode easily. For this reason, gold is most often used in plating where it is exposed to the air.Is wood a good conductor of electricity?
Wood, especially dry wood, is an insulator. An insulator prevents the flow of electricity while conductors allow electricity flow. The size of the material and temperature also affect the ability of a material to become an insulator or a conductor.What is the best insulator?
A: The best insulator in the world right now is most probably aerogel, with silica aerogels having thermal conductivities of less than 0.03 W/m*K in atmosphere. of aerogel preventing ice from melting on a hot plate at 80 degrees Celsius! Aerogel has its amazing properties because it's mostly made out of air.Which metal is not conductive?
The common metals that have the highest resistivity (lowest conductivity) are: Mercury. Stainless steel varieties. Titanium.What are conductive objects?
A conductor is a material which gives very little resistance to the flow of an electric current or thermal energy. Metals are the most conductive and insulators (ceramics, wood, plastics) the least conductive. Electrical conductivity tells us how well a material will allow electricity to travel through it.What is non conductive?
1. nonconductive - not able to conduct heat or electricity or sound. nonconducting, non-conducting. conductive - having the quality or power of conducting heat or electricity or sound; exhibiting conductivity.Is there a metal that does not conduct heat?
The poorest conductor of heat among metals is Bismuth. Stainless steel is another one that is a poor conductor of heat, and you use this often in everyday life! Other poor conductors include titanium, lead and chromium. And most ironically, Mercury, the liquid metal used in thermometers!Is carbon a good conductor of electricity?
The form of carbon that is a good conductor of electricity is Graphite. Graphite is an allotrope of carbon that has the properties of both metal and non metal. The reason why graphite is a good conductor of electricity is because of the mobility of the electrons in its outer valence shell.