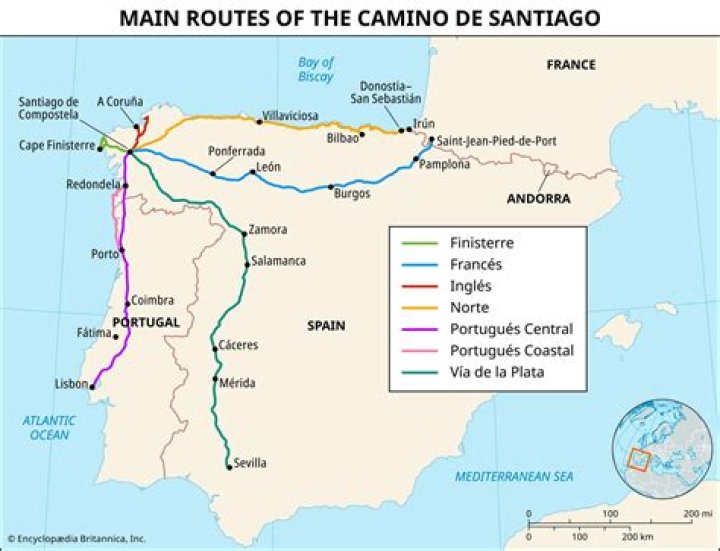
What is the formula for calculating heat?
What is the formula for calculating heat?
The heat capacity and the specific heat are related by C=cm or c=C/m. The mass m, specific heat c, change in temperature ΔT, and heat added (or subtracted) Q are related by the equation: Q=mcΔT.
How do you calculate heat and temperature?
The quantitative relationship between heat transfer and temperature change contains all three factors: Q = mcΔT, where Q is the symbol for heat transfer, m is the mass of the substance, and ΔT is the change in temperature. The symbol c stands for specific heat and depends on the material and phase.
What is Q in Q MC ∆ T?
Q = mc∆T. Q = heat energy (Joules, J) m = mass of a substance (kg) c = specific heat (units J/kg∙K) ∆ is a symbol meaning “the change in”
What is heat equation in PDE?
The heat equation is a consequence of Fourier’s law of conduction (see heat conduction). If the medium is not the whole space, in order to solve the heat equation uniquely we also need to specify boundary conditions for u. The heat equation is the prototypical example of a parabolic partial differential equation.
Who discovered the heat equation?
Jean Baptiste Joseph Fourier
In the early 19th century, while the study of thermodynamics was still in its infancy, the French scientist Jean Baptiste Joseph Fourier presented a remarkable formula describing the conduction of heat in a solid. In 1807, when Fourier presented this work, opinions were still divided about the nature of heat.
What is the difference between temperature and heat?
Heat measures how energy moves or flows. Temperature describes the average kinetic energy of molecules within a material or system and is measured in Celsius (°C), Kelvin(K), Fahrenheit (°F), or Rankine (R).
What does C stand for in Q mC?
Specific heat
Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K) ∆T = Change in temperature (Kelvins, K)
What is the L in Q mL?
specific latent heat
The specific latent heat (L) of a material… is a measure of the heat energy (Q) per mass (m) released or absorbed during a phase change. is defined through the formula Q = mL. is often just called the “latent heat” of the material.
What is Laplace equation for heat flow?
The solutions of Laplace’s equation are the harmonic functions, which are important in multiple branches of physics, notably electrostatics, gravitation, and fluid dynamics. In the study of heat conduction, the Laplace equation is the steady-state heat equation.
Is heat an elliptic equation?
The steady state heat equation (also called the steady state heat conduction equation) is elliptic whether it is 2-D or 3-D. The unsteady heat conduction equation (in 1-D, 2-D or 3-D) is parabolic in time.
What is another name of heat equation?
What is another name for heat equation? Explanation: The heat equation is also known as the diffusion equation and it describes a time-varying evolution of a function u(x, t) given its initial distribution u(x, 0). 6. Heat Equation is an example of elliptical partial differential equation.
What is the formula to find the amount of heat?
The formula is: Cp = Q/mΔT . You can manipulate this formula if you want to find the change in the amount of heat instead of the specific heat. Here’s what it would look like:
What is the equation for finding initial temperature?
where Q is the amount of heat energy added, m is the substance’s mass, c is specific heat, a constant, and ΔT means “change in temperature.” Make sure your units of measurement match the units used in the specific heat constant! For example, sometimes the specific heat may use Celsius.
How to calculate the specific heat of a material?
Study the equation. First, you should look at the equation to get a sense of what you need to do to find the specific heat. Let’s look at this problem: Find the specific heat of 350 g of an unknown material when 34,700 Joules of heat are applied, and the temperature rises from 22ºC to 173ºC with no phase change.
Do you need to know math to calculate heat load?
Don’t worry though. You won’t need to be a math prodigy to determine heat load. By reworking the heat load equation, we get m = Q/ ( Cp ×ΔT). That doesn’t help us much, since we are looking for the mass flow rate so we can enter it into our heat load equation.
What must equations be used to calculate heat?
To calculate the amount of heat released in a chemical reaction, use the equation Q = mc ΔT, where Q is the heat energy transferred (in joules), m is the mass of the liquid being heated (in kilograms), c is the specific heat capacity of the liquid (joule per kilogram degrees Celsius), and ΔT is the change in temperature of the liquid (degrees Celsius).
What is the formula to calculate specific heat?
Learn the equation for specific heat. Once you become familiar with the terms used for calculating specific heat, you should learn the equation for finding the specific heat of a substance. The formula is: C p = Q/mΔT. You can manipulate this formula if you want to find the change in the amount of heat instead of the specific heat. Jun 21 2019
What are the units in the heat equation?
Heat (or thermal) energy of a body with uniform properties: Heat energy = cmu, where m is the body mass, u is the temperature, c is the specific heat, units [c] = L2T−2U−1 (basic units are M mass, L length, T time, U temperature). c is the energy required to raise a unit mass of the substance 1 unit in temperature.
What is the heat equation formula?
The formula for specific heat looks like this: c = Q / (m * ΔT) Q is the amount of supplied or subtracted heat (in joules), m is the mass of the sample and ΔT is the difference between the initial and final temperature of the sample. Heat capacity is measured in J/(kg * K).