What is the enthalpy of combustion of butane?
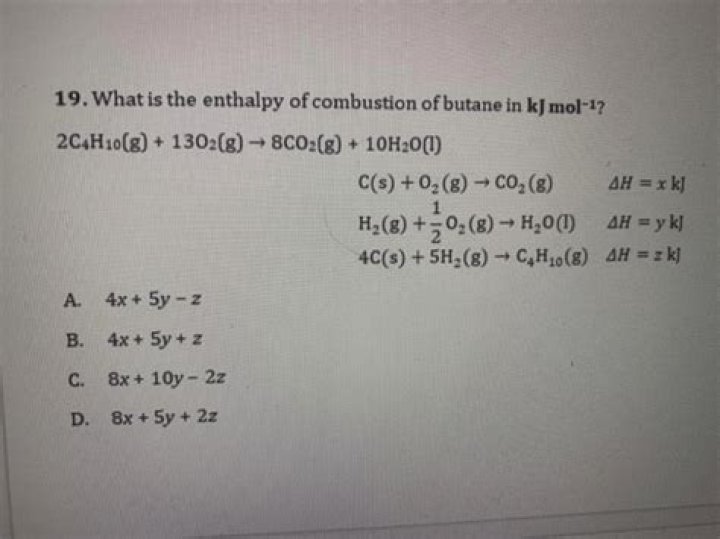
What is the enthalpy of combustion of butane?
2877.5 kJ/mol
Thermodynamic properties
| Phase behavior | |
|---|---|
| Gas properties | |
| Std enthalpy change of formation, ΔfHogas | –124.7 kJ/mol |
| Standard molar entropy, Sogas | 310.23 J/(mol K) |
| Enthalpy of combustion, ΔcHo | –2877.5 kJ/mol |
What is the excess reactant for a butane lighter?
“3.69 g” of butane reacts completely with an excess of oxygen gas to produce carbon dioxide and water.
What is the standard enthalpy of formation of butane C4H10 given the data below?
The enthalpy of formation of butane is −126 kJ/mol.
How do you calculate the combustion of butane?
C4H10 + O2 → CO2 + H2O .
What is the enthalpy of combustion for octane?
Enthalpy of combustion of liquid at standard conditions (nominally 298.15 K, 1 atm.)
| ΔcH°liquid (kJ/mol) | -5430. ± 100. |
| Method | AVG |
| Reference | N/A |
| Comment | |
|---|---|
| Average of 6 values; Individual data points |
What is the word equation for combustion of butane?
The butane combustion equation is: 2C4 H10 + 13O2 → 8CO2 + 10H2 O.
Why are Hess cycles needed?
Hess’ law can be used to determine the overall energy required for a chemical reaction, when it can be divided into synthetic steps that are individually easier to characterize. This affords the compilation of standard enthalpies of formation, that may be used as a basis to design complex syntheses.
How do you calculate the enthalpy of combustion?
Explanation:
- The standard enthalpy of combustion is ΔH∘c .
- You calculate ΔH∘c from standard enthalpies of formation:
- For each product, you multiply its ΔH∘f by its coefficient in the balanced equation and add them together.
How do you calculate heat of combustion?
Find the amount of substance burned by subtracting the final mass from the initial mass of the substance in g. Divide q in kJ by the mass of the substance burned. The answer is the experimental heat of combustion in kJ/g.

