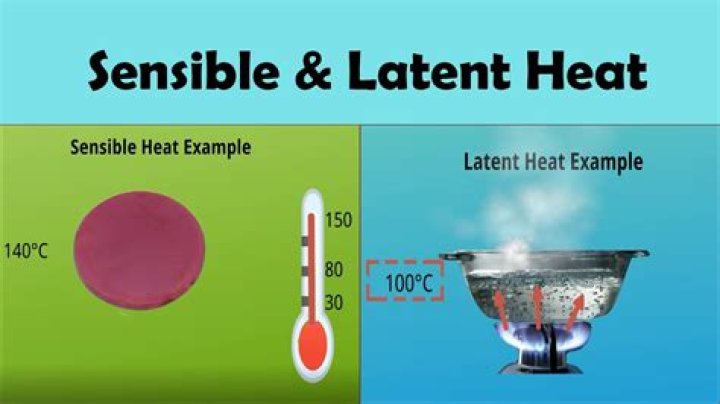
Latent and sensible heat are types of energy released or absorbed in the atmosphere. Latent heat is related to changes in phase between liquids, gases, and solids. Sensible heat is related to changes in temperature of a gas or object with no change in phase..
In this manner, what does sensible heat mean?
Definition of sensible heat. : thermal energy whose transfer to or from a substance results in a change of temperature — compare latent heat.
Furthermore, can sensible heat be measured? Sensible heat can be sensed, or measured, with a thermometer, and the addition or removal of sensible heat will always cause a change in the temperature of the substance. LATENT HEAT is the heat absorbed or given off by a substance while it is changing its physical state.
Furthermore, what is the difference between sensible heat and specific heat?
Heat that results in a temperature change is said to be "sensible" (although this term is falling out of favor). The specific heat capacity (which is often shortened to specific heat ) of a material is the amount of heat required to change a unit mass of a substance by one unit of temperature.
What is the difference between sensible and total cooling capacity?
The entire sum of cooling capacity generated is known as the total cooling capacity. The proportion that is used for purposely cooling the air is called sensible cooling capacity. Any proportion of the cooling capacity inadvertently used to dehumidify the air is called latent cooling capacity.
Related Question Answers
What is an example of latent heat?
Simple examples of latent heat: When we boil the water and the temperature remains at 100°C until the last drop evaporates, due to the latent heat of vaporization, the added heat in the water is absorbed and carried away by releasing vapor molecules. Latent heat of fusion of water: That is 334kJ/kg.What are the three types of heat transfer?
The three types of heat transfer Heat is transfered via solid material (conduction), liquids and gases (convection), and electromagnetical waves (radiation). Heat is usually transfered in a combination of these three types and seldomly occurs on its own.What is the total heat?
Noun. 1. total heat - (thermodynamics) a thermodynamic quantity equal to the internal energy of a system plus the product of its volume and pressure; "enthalpy is the amount of energy in a system capable of doing mechanical work" enthalpy, heat content, H.What is sensible heat formula?
As the name implies, sensible heat is the heat that you can feel. This is written as: Sensible heat = (mass of the body) * (specific heat capacity) * (change of the temperature) The equation is. Q= m c ΔT.What does latent heat cause?
Latent heat. All pure substances in nature are able to change their state. Solids can become liquids (ice to water) and liquids can become gases (water to vapor) but changes such as these require the addition or removal of heat. The heat that causes these changes is called latent heat.How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.What is the latent heat of water?
Latent heat of water is the energy required to change its state, also called its phase, without changing its temperature. The latent heat of fusion is the energy required to change a defined quantity of ice at 0 deg C from a solid to a liquid at the same temperature.What is latent heat transfer?
Latent Heat Transfer. At the point where one of the substances is ready to change state or phases (solid to liquid, liquid to gas, etc), heat is transferred from one substance without a corresponding temperature shift in the other substance.What is heat that Cannot be measured called?
The heat that cannot be measured by temperature likewise cannot be sensed by thermometer is called latent heat. Latent is the amount of heat that is absorbed into a substance in order to change its state.Which of the following is an example of sensible heating?
Sensible heat is literally the heat that can be felt. It is the energy moving from one system to another that changes the temperature rather than changing its phase. For example, it warms water rather than melting ice. In figure 2, very cold ice has heat added to it.What is meant by sensible heat?
Latent and sensible heat are types of energy released or absorbed in the atmosphere. Latent heat is related to changes in phase between liquids, gases, and solids. Sensible heat is related to changes in temperature of a gas or object with no change in phase.What is the unit of latent heat?
Latent heat is the amount of energy required to change the phase of 1 kg of a material and hence the unit is Joules per Kilogram.What is sensible load in HVAC?
The sensible cooling load refers to the dry bulb temperature of the building and the latent cooling load refers to the wet bulb temperature of the building. For summer conditions the humidity influence on the selection of the HVAC equipment and the latent load as well as the sensible load must be calculated.What is the specific heat of water?
The specific heat of water is 1 calorie/gram °C = 4.186 joule/gram °C which is higher than any other common substance. As a result, water plays a very important role in temperature regulation.What is subcooling in HVAC?
The term subcooling also called undercooling refers to a liquid existing at a temperature below its normal boiling point. A subcooled liquid is the convenient state in which, say, refrigerants may undergo the remaining stages of a refrigeration cycle.How can heat be measured?
Measurement of heat is done in calories. One calorie is the amount of energy required to raise one gram of water one degree Celsius. To measure heat, you divide the change in temperature of a sample of water by the mass of the water.What is the study of heat flow?
Thermodynamics is the study of heat energy and other types of energy, such as work, and the various ways energy is transferred within chemical systems.What is the instrument for measuring heat?
Calorimeter
Is heat of vaporization equal to heat of condensation?
The heat of vaporization of water is about 2,260 kJ/kg, which is equal to 40.8 kJ/mol. The vaporization is the opposite process of condensation. The heat of condensation is defined as the heat released when one mole of the substance condenses at its boiling point under standard pressure.