The current from the power source pushes electrons on to the electrode on the right of the diagram, where they cause reduction of species - hence this electrode is the cathode. In electrolytic cells, the cathode is negatively charged. Electrons are pushed on to the cathode by the external power source..
Furthermore, what is the cathode in an electrochemical cell?
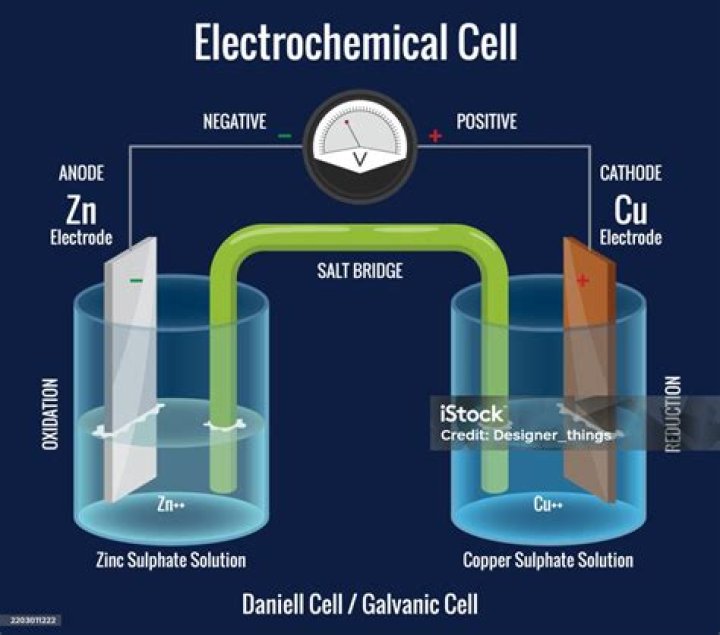
In both kinds of electrochemical cells, the anode is the electrode at which the oxidation half-reaction occurs, and the cathode is the electrode at which the reduction half-reaction occurs.
Additionally, is the cathode positive or negative? In a galvanic (voltaic) cell, the anode is considered negative and the cathode is considered positive. This seems reasonable as the anode is the source of electrons and cathode is where the electrons flow. However, in an electrolytic cell, the anode is taken to be positive while the cathode is now negative.
People also ask, what is the charge on cathode?
The cathode is the negatively charged electrode. The cathode attracts cations or positive charge. The cathode is the source of electrons or an electron donor. It may accept positive charge.
Why cathode is positive in electrochemical cell?
The anode is the electrode where oxidation (loss of electrons) takes place; in a galvanic cell, it is the negative electrode, as when oxidation occurs, electrons are left behind on the electrode. This is why the cathode is a positive electrode; because positive ions are reduced to metal atoms there.
Related Question Answers
What is the difference between anode and cathode?
Difference Between Anode And Cathode Here are some of the key differences between cathode and anode. The anode is the electrode where electricity moves into. The cathode is the electrode where electricity is given out or flows out of. In an electrolytic cell, a reduction reaction takes place at the cathode.Why DC current is used in electrolysis?
Direct current (DC) is used for electrolysis. The direct current helps to deposit the anions in the anode and the cations in the cathode. If alternate current was used, then the direction of current will go on changing and hence this would lead to uneven deposition of ions in the electrodes.What happens at the cathode?
Explanation: At the cathode in an electrolytic cell, ions in the surrounding solution are reduced into atoms, which precipitate or plate out on to the solid cathode. The anode is where oxidation takes place, and the cathode is where reduction takes place.Why anode is negatively charged?
Explanation: In a galvanic cell, electrons will move in to the anode. Since electrons carry a negative charge, then the anode is negatively charged. It's because the protons are attracted to the cathode, so it's mainly positive, and therefore is positively charged.How does anode and cathode work?
The cathode and anode (the positive and negative sides at either end of a traditional battery) are hooked up to an electrical circuit. The chemical reactions in the battery causes a build up of electrons at the anode. This results in an electrical difference between the anode and the cathode.What do you mean by anode?
An anode is the electrode in a polarized electrical device through which current flows in from an outside circuit. Cathodes get their name from cations (negatively charged ions) and anodes from anions (positively charged ions). In a device that consumes electricity, the anode is the charged positive electrode.Is copper a cathode or anode?
To purify copper electrolytically, the impure copper metal is made the anode (the positive electrode) in an electrolytic cell. A thin sheet of previously purified copper is used as the cathode (the negative electrode).What is an example of a cathode?
For example, reversing the current direction in a Daniell galvanic cell converts it into an electrolytic cell where the copper electrode is the positive terminal and also the anode. In a diode, the cathode is the negative terminal at the pointed end of the arrow symbol, where current flows out of the device.What is difference between cation and cathode?
#cation is a positively charged ion,it should attract to cathode. The main difference in cathode and cation is: #cathode is a negatively charged electrode by which electrons enter to an electric device. #cation is a positively charged ion,it should attract to cathode.What is produced at the cathode?
The electrolysis of other compounds All ionic compounds when molten can be decomposed when electricity is passed through using electrolysis. The metal and hydrogen always forms at the cathode. Non-metal always forms at the anode. Cations travel to the cathode.What are the materials used for anode and cathode?
Desirable properties for anode, cathode, and electrolyte materials are noted below. Metals such as Zinc and Lithium are often used as anode materials.Why is the cathode negative?
If the cathode has its electrons pulled from it by an atom or molecule or ion or thing, then the cathode becomes positively charged. This happens in an AA battery. If electrons are pumped onto the cathode and those electrons force a chemical reaction or reactions to occur, then the cathode is negative.What is charge of anode?
In electronic vacuum devices such as a cathode ray tube, the anode is the positively charged electron collector. In a tube, the anode is a charged positive plate that collects the electrons emitted by the cathode through electric attraction. It also accelerates the flow of these electrons.Is anion positive or negative?
Anion vs. Cation. Ions result from atoms or molecules that have gained or lost one or more valence electrons, giving them a positive or negative charge. Those with a negative charge are called anions and those with a positive charge are called cations.How do you make anode and cathode?
Use a gel that consists primarily of zinc powder for the anode of the battery. Separate the cathode and anode with a layer of paper and place them in a metal container. Seal the container to make the battery. Include electrodes as part of a secondary cell, such as a rechargeable battery.Why do electrons flow from anode to cathode?
By definition, a cathode is a negatively charged electrode (a metal plate or a wire), and an anode is a positively charged electrode. Therefore, electrons are repelled by the cathode and are attracted to the anode, which results in the current of electrons flowing from the cathode to the anode.Is anode red or black?
Anode: In tDCS, the positive (+) “stimulating” electrode, usually indicated with the color red. Bilateral: Relating to or affecting both sides. Cathodal: A descriptive term referring to the Cathode (negative, black) electrode. Cathode: In tDCS, the negative (-) electrode, usually indicated with the color black.How does an anode work?
Sacrificial anodes are used to protect metal structures from corroding. Sacrificial anodes work by oxidizing more quickly than the metal it is protecting, being consumed completely before the other metal reacts with the electrolytes. Three metals that can be used as sacrificial anodes are zinc, aluminum, and magnesium.Does electricity flow from anode to cathode?
Flow of Electrons Electrons always flow from the anode to the cathode or from the oxidation half cell to the reduction half cell. In terms of Eocell of the half reactions, the electrons will flow from the more negative half reaction to the more positive half reaction.