What is the charge of lanthanum
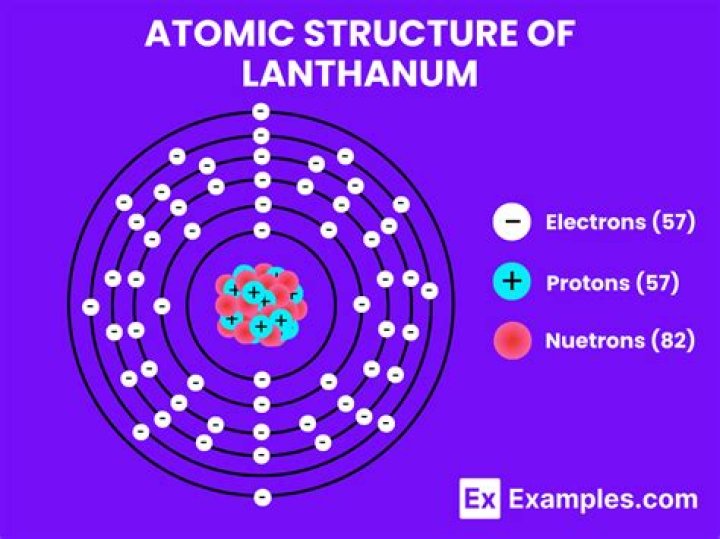
Lanthanum is traditionally counted among the rare earth elements. The usual oxidation state is +3.
Is lanthanum an ion?
Lanthanum Cation (3) is the ionized form of lanthanum (trivalent ion) with phosphate binding property. Lanthanum ion binds dietary phosphate and inhibits the absorption of phosphate by forming highly insoluble lanthanum-phosphate complexes that reduce the concentrations of serum phosphate and calcium phosphate.
What is the charge of actinium?
Quantityion charge: +3, coordination number: 6112 pmPauling Empirical Crystal Radiusion charge: +3118 pm
What is lanthanum?
Lanthanum is also found in the anode of nickel metal hydride batteries used in hybrid cars. Lanthanum is an important component of mischmetal alloy (about 20%). … ‘Rare earth’ compounds containing lanthanum are used extensively in carbon lighting applications, such as studio lighting and cinema projection.What is ND in chemistry?
neodymium (Nd), chemical element, a rare-earth metal of the lanthanide series of the periodic table.
What is mn on the periodic table?
manganese (Mn), chemical element, one of the silvery white, hard, brittle metals of Group 7 (VIIb) of the periodic table.
Where is in on the periodic table?
Element NameSymbolAtomic NumberHydrogenH1IndiumIn49IodineI53IridiumIr77
Is lanthanum metallic or nonmetallic?
Lanthanum is a ductile and malleable silvery white metal that is soft enough to be cut with a knife. It is the second most reactive of the rare-earth metals after europium.What is lanthanum made of?
Lanthanum is found in rare-earth minerals such as cerite, monazite, allanite, and bastnasite. Monazite and bastnasite are principal ores in which lanthanum occurs in percentages up to 25 percent and 38 percent respectively. Misch metal, used in making lighter flints, contains about 25 percent lanthanum.
What is 230 on the periodic table?Thorium-230 | Th – PubChem.
Article first time published onWhat is the charge of the nucleus of 89?
Element 89 has an atomic number of 89 in the periodic table. … The number indicates that every nucleus of an actinium atom has 89 protons. Therefore, the charge of an actinium atom nucleus must be +89.
Is neptunium a metal nonmetal or metalloid?
Neptunium is a hard, silvery, ductile, radioactive actinide metal. In the periodic table, it is located to the right of the actinide uranium, to the left of the actinide plutonium and below the lanthanide promethium. Neptunium is a hard metal, having a bulk modulus of 118 GPa, comparable to that of manganese.
How do you write electronic configuration?
Writing Electron Configurations. When writing an electron configuration, first write the energy level (the period), then the subshell to be filled and the superscript, which is the number of electrons in that subshell. The total number of electrons is the atomic number, Z.
Why is lanthanum 5d1?
In terms of the electrons they are most stable in the ground state or lowest possible energy. … By the addition of one electron to the 5d raises the energy of the level so that the next electrons go to 4f. Hence Lanthanum [Xe] 5d1 6s2 instead of [Xe] 6s2 4f1.
How much Neodymium is left in the world?
Reserves of neodymium are estimated to be 8 million tonnes, world production of neodymium oxide is about 7.000 tonnes a year.
Is PM a metal?
PromethiumAppearancemetallicMass number[145]Promethium in the periodic table
What group does ND belong to on periodic table?
NeodymiumGroupgroup n/aPeriodperiod 6Blockf-blockElectron configuration[Xe] 4f4 6s2
What are the numbers on the periodic table?
The number above the symbol is the atomic mass (or atomic weight). This is the total number of protons and neutrons in an atom. The number below the symbol is the atomic number and this reflects the number of protons in the nucleus of each element’s atom.
How do you find number of electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
What type of solid is mn?
Atomic Mass54.93804uElectron Configuration[Ar]4s23d5Oxidation States+7, +4, +3, +2Year Discovered1774
What is the oxidation state of Mn in MnO?
The oxidation number of Mn in [MnO4]− ion is +7.
Who is modern periodic table?
In the 1860s, a scientist named Dmitri Mendeleev also saw the need to organize the elements. He created a table in which he arranged all of the elements by increasing atomic mass from left to right across each row.
What is 59 on the periodic table?
Praseodymium is a chemical element with symbol Pr and atomic number 59. Classified as a lanthanide, Praseodymium is a solid at room temperature.
What does a stand for in the periodic table?
The chemical elements of the periodic chart sorted by:SymbolName chemical element- ElectronegativityArArgon- DensityAsArsenic- Melting pointAtAstatine- Boiling pointAuGold
Is nickel a nonmetal?
nickel (Ni), chemical element, ferromagnetic metal of Group 10 (VIIIb) of the periodic table, markedly resistant to oxidation and corrosion.
How is lanthanum processed?
Today, lanthanum is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements that can contain as much as 25% lanthanum. … Lanthana (La2O3) is used to make the glass used in camera lenses and in other special glasses.
Is lanthanum a conductor?
Lanthanum has a relatively high resistivity of 615 nΩm at room temperature; in comparison, the value for the good conductor aluminium is only 26.50 nΩm. Lanthanum is the least volatile of the lanthanides.
Is thorium named after Thor?
Discovery date1829Origin of the nameThorium is named after Thor, the Scandinavian god of war.Allotropes
What is the half life of uranium?
The half-life of uranium-238 is about 4.5 billion years, uranium-235 about 700 million years, and uranium-234 about 25 thousand years.
What is 92 on the periodic table?
Uranium is a silvery-white metallic chemical element in the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium has the highest atomic weight (19 kg m) of all naturally occurring elements.
What is a charge of a nucleus?
The nucleus has an overall positive charge as it contains the protons. Every atom has no overall charge (neutral). This is because they contain equal numbers of positive protons and negative electrons. These opposite charges cancel each other out making the atom neutral.