What is specific heat a measure of?
What is specific heat a measure of?
specific heat, the quantity of heat required to raise the temperature of one gram of a substance by one Celsius degree. The units of specific heat are usually calories or joules per gram per Celsius degree.
What is the specific heat of a substance called?
The specific heat (also called specific heat capacity) is the amount of heat required to change a unit mass (or unit quantity, such as mole) of a substance by one degree in temperature. i.e., 1 calorie is needed per degree Kelvin (or Celsius) of temperature change for 1 gram of liquid water.
Is specific heat the same as joules?
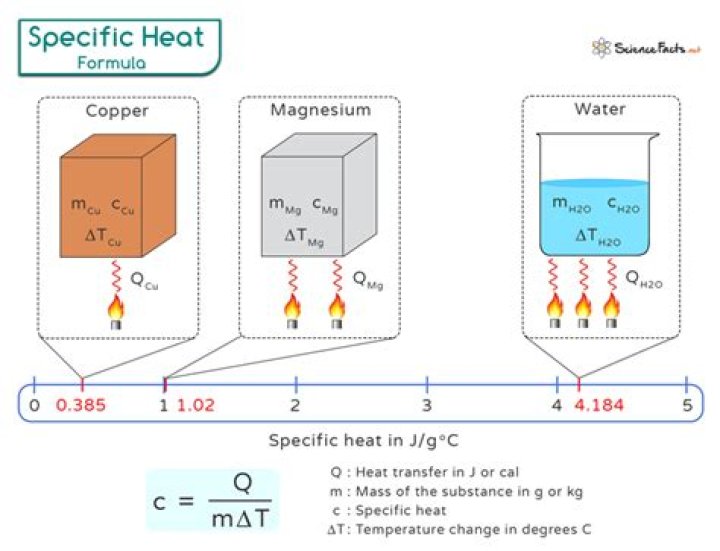
Specific heat is also sometimes referred to as massic heat capacity. The SI unit of specific heat capacity is joule per kelvin per kilogram, J⋅kg−1⋅K−1. For example, the heat required to raise the temperature of 1 kg of water by 1 K is 4184 joules, so the specific heat capacity of water is 4184 J⋅kg−1⋅K−1.
What is specific heat of a liquid?
For liquid at room temperature and pressure, the value of specific heat capacity (Cp) is approximately 4.2 J/g°C. This implies that it takes 4.2 joules of energy to raise 1 gram of water by 1 degree Celsius.
What does specific mean in specific heat capacity?
Specific heat capacity is the amount of heat energy required to raise the temperature of a substance per unit of mass. The specific heat capacity of a material is a physical property. It is also an example of an extensive property since its value is proportional to the size of the system being examined.
What is the specific heat of a substance apex?
The specific heat of a substance is the amount of energy required to raise the temperature of 1 gram of the substance by 1oC.
What is specific heat of liquid?
What is value of specific heat of water?
The SI unit of specific heat capacity is joule per kelvin per kilogram, J⋅kg−1⋅K−1. For example, the heat required to raise the temperature of 1 kg of water by 1 K is 4184 joules, so the specific heat capacity of water is 4184 J⋅kg−1⋅K−1.
What is high specific heat?
A high specific heat capacity means it can hold a large amount of thermal energy in for a low mass or temperature change. It is also good at keeping in thermal energy, for example: Object: Mass = 3kg.
What is specific heat of a substance class 12?
Specific heat is the amount of heat required to raise the temperature of 1 kg of the substance through 1oC.
What substances have high specific heat?
Water has the highest specific heat capacity of any liquid. Specific heat is defined as the amount of heat one gram of a substance must absorb or lose to change its temperature by one degree Celsius.

