What is photoluminescence spectrum?
What is photoluminescence spectrum?
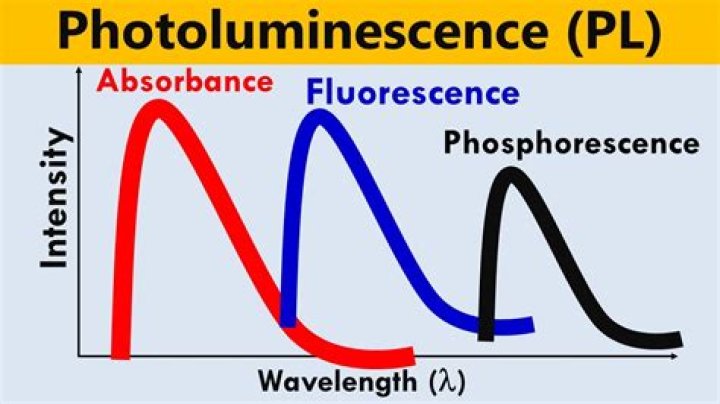
Photoluminescence spectroscopy, often referred to as PL, is when light energy, or photons, stimulate the emission of a photon from any matter. It is a non-contact, nondestructive method of probing materials. Photoluminescence used in Fluorescence spectroscopy can provide two results: Fluorescence and Phosphorescence.
How is photoluminescence spectra measured?
Photoluminescence spectra are recorded by measuring the intensity of emitted radiation as a function of either the excitation wavelength or the emission wavelength. An excitation spectrum is obtained by monitoring emission at a fixed wavelength while varying the excitation wavelength.
What does PL measure?
PL (Photoluminescence Spectroscopy) uses a laser beam to capture light generated from a substance as it falls from the excited state to ground state when irradiated by a laser beam. By measuring the luminescence spectrum, it is possible to observe material imperfections and impurities.
How do you determine the excitation wavelength for photoluminescence?
the range of excitation wavelength should be 200 nm to 20 nm less than your emission wavelength. 6. Now the last point, check the excitation spectra and find out the highest intense peak. The wavelength belongs to highest intense peak, is the suitable excitation wavelength for ur sample.
What is photoluminescence with example?
Clocks and watches have phosphorescent paint on them. Photoluminescence – This is a process where a substance absorbs photons and then re-emits them. The electromagnetic energy is absorbed at a certain wavelength and is emitted at a different wavelength which is most commonly longer.
What causes photoluminescence?
Photoluminescence is the emission of light which is caused by the irradiation of a substance with other light. The term embraces both fluorescence and phosphorescence, which differ in the time after irradiation over which the luminescence occurs.
What is PL excitation spectra?
Photoluminescence (PL) spectroscopy is a form of light emission spectroscopy in which the light emission comes from a process called photo-excitation. As the light is directed onto a sample, the electrons within the material move into excited states. (
What is PL mapping?
Gallium Nitride (GaN) nanowires mapping Photoluminescence (or PL) and Raman are spectroscopy techniques providing information on optical and electrical properties of semiconductors and many nano-materials. The nanowire is 150nm in diameter.
What is the difference between fluorescence emission spectrum and fluorescence excitation spectrum?
What would be the difference between an excitation and emission spectrum in fluorescence spectroscopy? In an emission spectrum, the excitation monochromator is set to some wavelength known to excite the sample and the emission monochromator is scanned through the different wavelengths.
What is emission and excitation spectra?
An emission spectrum describes the wavelengths of the spectrum emitted by an energetic object. The excitation spectrum is a range of light wavelengths that add energy to a fluorochrome, causing it to emit wavelengths of light, the emission spectrum2.

