What is nh4po4? | ContextResponse.com
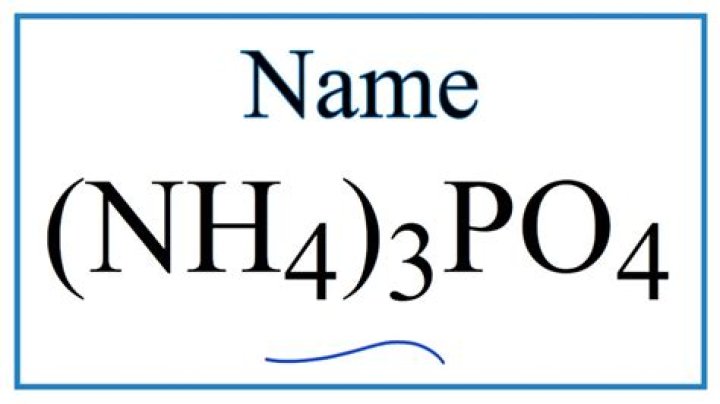
.
Beside this, what is ammonium phosphate used for?
Ammonium phosphate is used as an ingredient in some fertilizers as a high source of elemental nitrogen. It is also used as a flame retardant in thermoplastic compositions.
Additionally, how is ammonium phosphate produced? Triammonium phosphate can be prepared in the laboratory by treating 85% phosphoric acid with 30% ammonia solution: H3PO4 + 3 NH3 → (NH4)3PO. The solid, which has the odor of ammonia, is readily soluble in water. The salt converts to diammonium hydrogen phosphate (NH4)2HPO4.
In respect to this, is ammonium phosphate dangerous?
INHALATION: if exposed to ammonia fumes from diammonium phosphate, give artificial respiration and oxygen if needed; enforce rest. EYES: flush with water for at least 15 min.; if irritation persists, get medical attention.
What is ammophos?
Mono ammonium phosphate (MAP or ammophos) is a complex, highly concentrated, ballastless, water-soluble mineral fertilizer. This fertilizer is input with the main treatment of the soil or prior to sowing.
Related Question AnswersWhat is DAP made of?
Diammonium phosphate (DAP) is the world's most widely used phosphorus fertilizer. It's made from two common constituents in the fertilizer industry, and its relatively high nutrient content and excellent physical properties make it a popular choice in farming and other industries.Is ammonium phosphate an acid or base?
Ammonium phosphate, dibasic is an instance of the compound where there are two replaceable hydrogen atoms, whereas the monobasic example has only one hydrogen atom to donate to the base within the acid-base reaction of the ammonium and phosphate.How do you make monoammonium phosphate?
Growing Monoammonium Phosphate Crystals- Stir six tablespoons of monoammonium phosphate into 1/2 cup of very hot water in a clear container.
- Add food coloring, if desired.
- Stir until the powder is completely dissolved.
Is ammonium phosphate a fertilizer?
Ammonium phosphate (16-20-0) adds nitrogen and phosphates to lawns that are lacking in those nutrients. Ammonium phosphate is a fast release fertilizer and can be used when planting new grass, for maintenance, when overseeding or when renovating the lawn.What is the difference between MAP and DAP fertilizer?
MAP is manufactured by combining one mole (molecular weight) of ammonia with one mole of phosphoric acid. DAP is produced by adding 2 moles of ammonia with one mole of phosphoric acid. The additional ammonia in DAP adds beneficial nitrogen, but can create unfavorable chemical reactions in soil solution.What is ammonium phosphate made of?
Ammonium phosphates are inorganic salts derived from the reaction between ammonia and phosphoric acid. They are produced as solids and fluids and they may contain phosphate values in ortho- (usually solid) and/or polyphosphate (usually fluid) form.What type of solid is ammonium phosphate?
Ammonium Phosphate. ( N H 4 ) 3 P O 4 (NH_4)_3PO_4 (NH4)3PO4(Ammonium phosphate ) is a ionic solid.it is a ionic solid as the constituent particles in the solid are ions and the constituent ions are N H 4 + , P O 4 3 − NH_4^{+},PO_4^{3-} NH4+,PO43− ions.Is ammonium a metal?
Ammonium metal Under normal conditions, ammonium does not exist as a pure metal, but does as an amalgam (alloy with mercury).Can fire extinguisher powder kill you?
It won't hurt you in small amounts and is quite safe to deal with. Fire extinguisher powder inhalation is one of the biggest dangers with fire extinguishers. It is terribly irritating to mucose membranes and will cause difficulties with respiration if indrawn in giant enough quantities.What is monoammonium phosphate used for?
Monoammonium phosphate (MAP) is a widely used source of phosphorus (P) and nitrogen (N).* It's made of two constituents common in the fertilizer industry and contains the most phosphorus of any common solid fertilizer.Is Ammonium Phosphate an electrolyte?
Ammonium Phosphate (NH4)3PO4 is a strong electrolyte.Is monoammonium phosphate toxic?
MAY CAUSE SKIN, EYE AND RESPIRATORY TRACT IRRITATION. Acute Inhalation: Dusts may cause upper respiratory tract irritation. Acute Ingestion: May cause abdominal cramps, nausea, vomiting, diarrhea. Skin Exposure: In case of contact, wash with plenty of soap and water.Why is ammonium phosphate a good Fertiliser?
Fertilisers may contain nitrogen, phosphorus and potassium compounds to promote plant growth. ammonium ions, NH 4 +, and nitrate ions, NO 3 -, are sources of soluble nitrogen. phosphate ions, PO 4 3-, are a source of soluble phosphorus. all common potassium compounds dissolve in water to produce potassium ions, K.Is ca3 po4 2 soluble in water?
Tricalcium phosphate (sometimes abbreviated TCP) is a calcium salt of phosphoric acid with the chemical formula Ca3(PO4)2.Tricalcium phosphate.
| Names | |
|---|---|
| Melting point | Liquifies under high pressure at 1670 K (1391 °C) |
| Solubility in water | 0.002 g/100 g |
| Thermochemistry | |

