What is a resonance structure simple definition?
What is a resonance structure simple definition?
Resonance structures are a set of two or more Lewis Structures that collectively describe the electronic bonding a single polyatomic species including fractional bonds and fractional charges.
What are the two types of resonance?
There are two types of Resonance effects namely positive resonance effect and negative resonance effect. Positive Resonance Effect- Positive resonance effect occurs when the groups release electrons to the other molecules by the process of delocalization.
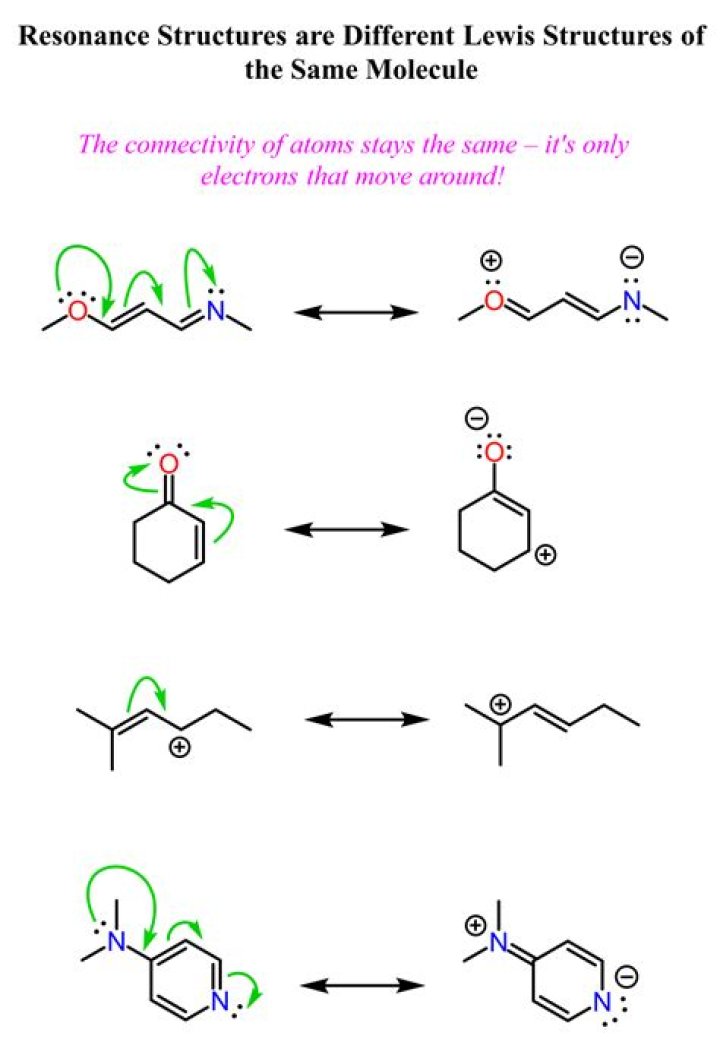
What does the arrow mean in resonance structure?
Every Resonance Form For A Molecule Can Be “Found” Through The Application Of Three Electron-Pushing Arrow “Moves” The charges change at the tail, which becomes more positive (since it’s giving away electrons), and the head, which becomes more negative (since it’s gaining electrons).
What does a resonance structure tell you?
Resonance is a mental exercise and method within the Valence Bond Theory of bonding that describes the delocalization of electrons within molecules. It compares and contrasts two or more possible Lewis structures that can represent a particular molecule.
What is resonance in chemistry class 12?
Hint: Resonance basically defines the polarity produced in a molecule by the interaction between a lone pair and a pi bond or the interaction of two pi bonds in adjacent atoms.
What is resonance and its types?
Resonance phenomena occur with all types of vibrations or waves: there is mechanical resonance, acoustic resonance, electromagnetic resonance, nuclear magnetic resonance (NMR), electron spin resonance (ESR) and resonance of quantum wave functions.
What are the types of resonance effect?
Two types of Resonance effects exist, namely:
- Positive resonance effect.
- Negative resonance effect.
What does a curved arrows represent?
Curved arrows represent electron movements. Chemists utilize curved-arrow (electron-pushing) notation as a bookkeeping tool to keep track of electron movements (bond breaking and formation) within and between compounds.
What do resonance structures represent about a molecule?
Resonance is the use of two or more Lewis structures to represent the covalent bonding in a molecule. One of the valid structures is referred to as a resonance structure.
How do resonance structures differ?
Resonance structures have the same number of electrons and therefore have the same overall charge. Resonance structures differ only in the arrangement of electrons; the atoms keep the same connectivity and arrangement.

