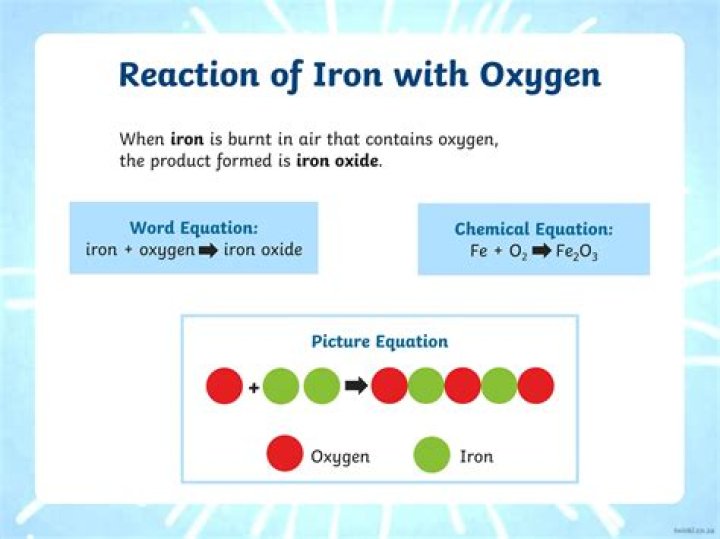
Iron reacts with water in the form of steam to form iron oxide, along with the release of hydrogen. It displaces hydrogen from water/steam, which is evolved or released as a gas..
Also question is, does Iron react vigorously with water?
Zinc and Iron on Water It reacts slowly with hot water to give the slightly soluble magnesium hydroxide (?? ??) and hydrogen.
Secondly, what happens when lead reacts with water? Reaction of lead with water The surface of metallic lead is protected by a thin layer of lead oxide, PbO. It does not react with water under normal conditions.
Also Know, what happens when iron reacts with water and oxygen?
Rust is an iron oxide, a usually red oxide formed by the redox reaction of iron andoxygen in the presence of water or air moisture. Several forms of rust are distinguishable both visually and by spectroscopy, and form under different circumstances.
What is the result of iron oxygen water?
Hope this helps you! Explanation: This above equation is the rusting process. When the iron comes in contact with oxygen along with water, it ends up forming ferric oxide, otherwise known as the rust.
Related Question Answers
Does Iron react with water?
Reaction of iron with water Air-free water has little effect upon iron metal. However, iron metal reacts in moist air by oxidation to give a hydrated iron oxide.What chemicals react violently with water?
Water-sensitive chemicals are those that react violently with water. The alkali metals such as sodium, potassium and lithium react with water to produce heat and flammable hydrogen gas, which can ignite or combine explosively with atmospheric oxygen.What metal catches fire in water?
Magnesium, lithium, sodium, potassium, caesium, and rubidium are all metals that will burn and react with water. Potassium is so reactive with water that it has to be stored in oil because it will react with the moisture in the air.What metal explodes in water?
Now this is an effective way for kids to remember the names of elements… have the elements explode! Five out of the six Alkali metals react with air and water: Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), and Caesium (Cs).How does iron react with cold water?
iron reacts with steam to give iron oxide and hydrogen gas. iron does not react with cold water but it reacts with hot water and steamWhat explodes when it touches water?
Sodium is one of the highly reactive alkali metals. In its pure form, the soft, shiny metal reacts in water to form sodium hydroxide and hydrogen gas.Which metal does not react with water?
Beryllium
Which alkali metal is most reactive?
Cesium
What is the formula for rust?
Fe2O3
Is Rust acidic or basic?
Metals are acidic. Rust is iron oxide. Metallic oxides are basic in nature. This means rust is base.What is Fe2O3 xH2O?
Fe2O3.xH2O is usually called hydrated ferric Oxide. It is a hydrated crystal. There are two different types of valency of Iron (Fe)— 2 and 3. When Ferrous ion forms an ionic bond with Oxygen, it is called Ferrous Oxide. And when with Ferric, it is Ferric Oxide.What does iron react with?
Iron is a very active metal. It readily combines with oxygen in moist air. The product of this reaction, iron oxide (Fe 2 O 3 ), is known as rust. Iron also reacts with very hot water and steam to produce hydrogen gas.What type of reaction is rusting of iron?
oxidation
What is the Fe2O3?
Iron(III) oxide or ferric oxide is the inorganic compound with the formula Fe2O3. As the mineral known as hematite, Fe2O3 is the main source of iron for the steel industry.What does iron do in blood?
Iron is an important component of hemoglobin, the substance in red blood cells that carries oxygen from your lungs to transport it throughout your body. Hemoglobin represents about two-thirds of the body's iron. If you don't have enough iron, your body can't make enough healthy oxygen-carrying red blood cells.What is the order of rusting of iron?
It occurs in moist air and in water. Rust requires three chemicals in order to form: iron, oxygen, and water. Because of the electrochemical nature of the reaction, dissolved electrolytes in water aid the reaction. Rust occurs more quickly in saltwater than in pure water, for example.What is the reaction of iron oxygen water?
During rusting, iron is oxidised by the oxygen of air in the presence of water (moisture) to form hydrated iron (III) oxide called rust. The electrolysis of water produces 2 volumes of hydrogen gas and 1 volume of oxygen gas. Hence, the ratio of hydrogen and oxygen elements in water is 2 : 1 by volume.Why is lead present in water?
Lead can enter drinking water when a chemical reaction occurs in plumbing materials that contain lead. This is known as corrosion – dissolving or wearing away of metal from the pipes and fixtures. This reaction is more severe when water has high acidity or low mineral content.Why lead does not react with water?
The surface of metallic lead is protected by a thin layer of lead oxide, PbO. Hence, it does not react with water under normal conditions.