What element has a charge of 35?
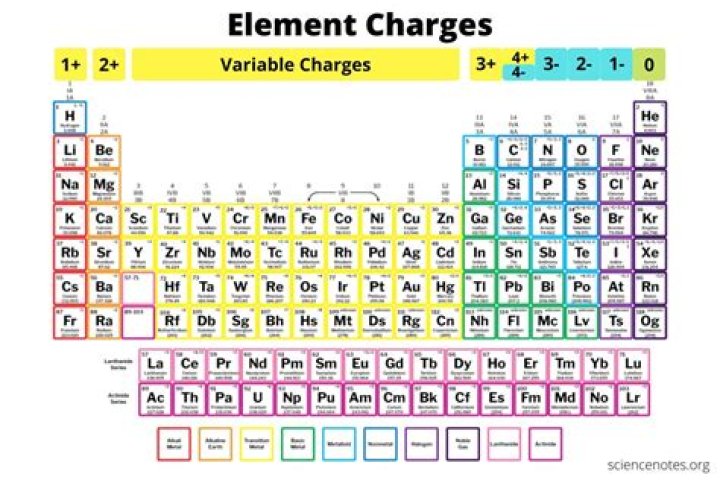
What element has a charge of 35?
bromine
Table of Common Element Charges
| Number | Element | Charge |
|---|---|---|
| 35 | bromine | 1-, 1+, 5+ |
| 36 | krypton | 0 |
| 37 | rubidium | 1+ |
| 38 | strontium | 2+ |
What element has 35 protons in its nucleus?
Bromine
| Name | Bromine |
|---|---|
| Atomic Number | 35 |
| Atomic Mass | 79.904 atomic mass units |
| Number of Protons | 35 |
| Number of Neutrons | 45 |
What element has a 1+ charge with 36 electrons?
bromine atom
For example, the neutral bromine atom, with 35 protons and 35 electrons, can gain one electron to provide it with 36 electrons. This results in an anion with 35 protons, 36 electrons, and a 1− charge. It has the same number of electrons as atoms of the next noble gas, krypton, and is symbolized Br−.
What element has 35 protons and 80 neutrons?
Bromine has 35 protons and a mass number of 80.
Which elements have a charge?
Carbon is a solid and oxygen is a gas; when they combine they become carbon dioxide a gas. Generally, metals on the Periodic Table of the Elements have a positive charge (a positive ion) and the nonmetals have a negative charge (a negative ion). There are several exceptions to this rule.
Why does pb have a 2+ charge?
If the elements in Group 4 form 2+ ions, they lose their p electrons, leaving the s2 pair unused. For example, to form a lead(II) ion, lead loses its two 6p electrons, but the 6s electrons are left unchanged, an “inert pair”. Pb+2 is “lead two ion”. …
Which element has 45 neutrons?
Rhodium –
#45 – Rhodium – Rh.
Is there an atomic mass of 41?
The Elements, sorted by Atomic Mass
| Atomic Number | Symbol | Name |
|---|---|---|
| 38 | Sr | Strontium |
| 39 | Y | Yttrium |
| 40 | Zr | Zirconium |
| 41 | Nb | Niobium |
How many electrons are in BR -?
35 electrons
The atomic number for bromine is 35, which means it has 35 protons in its atomic nuclei. A neutral bromine atom would also have 35 electrons.
How do I know the charge of an element?
How to Find the Charge of an Element
- Use the periodic table. The usual charge of an element is common to its group.
- Use a chart.
- For a single atom, the charge is the number of protons minus the number of electrons.
- Find the charge by balancing charge in a compound.
What elements have no charge at all?
neutron: A subatomic particle forming part of the nucleus of an atom. It has no charge. It is equal in mass to a proton or it weighs 1 amu.
Which is more stable Pb2+ and pb4+?
When ns2 electrons of outermost shell do not participate in bonding, it is called inert pair effect. Since the inert pair effect increases down the group, therefore Pb2+ compounds are more stable than Pb4+ compounds.
Which is the most stable charge of an element?
An atom of an element is most stable when its outer electron shell is completely filled or half-filled. The most common charges are based on maximum stability for the atom. However, other charges are possible.
Which is an element with a charge of zero?
Common Charges of Element Atoms. However, other charges are possible. For example, hydrogen sometimes has a charge of zero or (less commonly) -1. Although noble gas atoms almost always carry a charge of zero, these elements do form compounds, which means they can gain or lose electrons and carry a charge.
How to calculate the charges of an element?
Chart of Common Charges of Chemical Elements This is a chart of the most common charges for atoms of the chemical elements. Use this chart to predict if an atom can bond with another atom. This is a chart of the most common charges for atoms of the chemical elements. Use this chart to predict if an atom can bond with another atom. Menu Home
What kind of atom has 10 electrons and no net charge?
If the element has 10 electrons and has a net charge of 0 which makes it a neutral atom, the atom must have 10 protons. Elements are defined by the number of protons which is also equal to the atomic number. #10^+ =# Atomic Number 10. On the Periodic Table this is Neon.