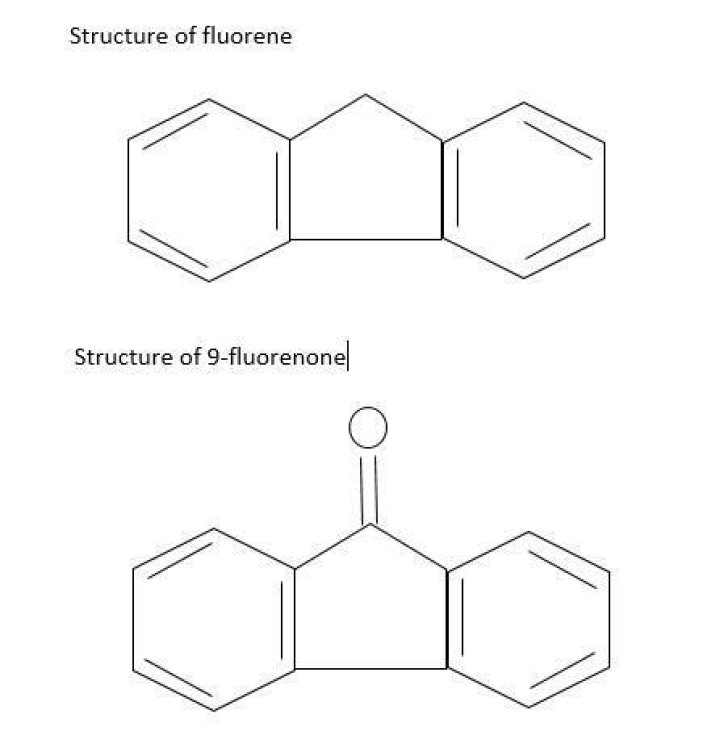
Fluorene /ˈfl??riːn/, or 9H-fluorene, is a polycyclic aromatic hydrocarbon. It forms white crystals that exhibit a characteristic, aromatic odor similar to that of naphthalene. It is combustible. It has a violet fluorescence, hence its name..
Simply so, is fluorene a mutagen?
Benzo[c]fluorene is a polycyclic aromatic hydrocarbon (PAH) with mutagenic activity. It is a component of coal tar, cigarette smoke and smog and thought to be a major contributor to its carcinogenic properties. According to the KEGG it is a group 3 carcinogen (not classifiable as to its carcinogenicity to humans).
Likewise, is fluorene polar or nonpolar? Structurally, fluorene has a five-carbon ring with a benzene ring on each side. Because carbon and hydrogen have electronegativity values that are not significantly different from one another, it is a non-polar molecule.
Keeping this in consideration, is fluorene an irritant?
* Fluorene can irritate and burn the eyes and skin. Fluorene is white crystalline plates. It is used in resinous products, dyestuffs, and as a chemical intermediate. * Fluorene is on the Hazardous Substance List because it is cited by HHAG and EPA.
What is fluorene used for?
Like most PAHs, fluorene is used to make dyes, plastics and pesticides. How can fluorene enter and leave your body? One of the most common ways fluorene can enter your body is through breathing contaminated air. It can get into your lungs when you breathe it.
Related Question Answers
Is fluorene an acid or base?
Acidity. The C9-H sites of the fluorene ring are weakly acidic (pKa = 22.6 in DMSO.) Deprotonation gives the stable fluorenyl anion, nominally C13H9−, which is aromatic and has an intense orange colour. The anion is a nucleophile.What is the structure of fluorene?
C13H10
What is the RF value of fluorene?
Fluorene is the least polar and moves very fast up the TLC plate. Fluorenone is in the middle for polarity and moves at a rate of 0.65, right in between fluorenol at 0.22 Rf and fluorene at 0.90 Rf value.Is 9 Fluorenone soluble in water?
9-fluorenone has one polar C=O. bond, but it also has two aromatic rings. Dispersion interactions are strong for such aromatic rings, and the point can be made that this factor will drive the solvation of 9-fluorenone in hexane. This is confirmed by the fact that 9-fluorenone is insoluble in water.What is the melting point of fluorene?
116 °C
Is Fluorenone soluble in water?
Fluorenone is soluble in a wide range of organic solvents (solvents that are carbon-hydrogen based) including things like chloroform, methanol, ethanol, dichloromethane, and acetonitrile. It is NOT, however, soluble in water. Overall, fluorenone is a polar compound, mainly due to the presence of the ketone.Is fluorene soluble in methanol?
You are correct. It's too soluble in toluene and insoluble in H2O so Methanol is your best choice. Bring the solvent to its boiling point (~65°C) and add just enough to barely cover the sample.Is 9 Fluorenone an acid or base?
9-fluorenone is not acidic or basic in water, and in insoluble in aqueous solutions. The benzoic acid was recovered on acidifying the basic extract. The ethyl 4- aminobenzoate was recovered by adding base to the acid extract.What is the melting point of 9 Fluorenol?
153-154 °C
Will fluorene or fluorenone elute first?
Fluorenone is more polar than Fluorene. Fluorene will elute off of your very polar Alumina column first (before Fluorenone) when using hexane as the eluting solvent.Why is 9 Fluorenone yellow?
The structure of 9-Fluorenone is as follows: When light pass through 9-fluorenone, keto group of 9-fluorenone absorbs light and there will be and transitions, and releases energy at visible region, due to these transitions wavelength of the light changes leads to color appearance.Is water Polar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.What color is 9 Fluorenol?
Fluorenol is an alcohol derivative of fluorene. In the most significant isomer, fluoren-9-ol or 9-hydroxyfluorene, the hydroxy group is located on the bridging carbon between the two benzene rings. Hydroxyfluorene can be converted to fluorenone by oxidation. It is a white-cream colored solid at room temperature.Are ethers polar?
Ethers are slightly polar. Ethers are more polar than alkenes but not as polar as alcohols, esters, or amides of comparable structure. The presence of two lone pairs of electrons on the oxygen atoms makes hydrogen bonding with water molecules possible.Why is 9 Fluorenone more polar than fluorene?
The solvent systems separated fluorene and 9-fluorenone based on their difference in structure and polarity. In principle, the chemical compound that flows through the column at a faster speed is more non-polar; therefore, in this case fluorene was more non-polar than 9-fluorenone.Why is dichloromethane polar?
Methylene chloride or dichloromethane is moderately polar. Any small molecule cannot be polar or non polar at the same time * polarity is a range. Chlorine being more electronegative than hydrogen causes the dichloromethane molecule to be slightly more negative on the side of the chlorine atoms.Is NaOH polar?
In alkali metal hydroxides, such as NaOH, the Na-O bond is most polar. The electronegativity difference between sodium and oxygen is larger than that between oxygen and hydrogen. A combination like that results in an ionic bond, not a covalent one, so sodium hydroxide should be thought of as Na+ and HO-.Why is methanol polar?
Methanol is a polar molecule: The alcohol (-OH) group dominates the molecule making it definitely polar. The electrostatic potential clearly shows that the oxygen is partially negative whereas the carbon and hydrogens are partially positive.Is fluorene organic?
Fluorene or 9H-fluorene is a polycyclic aromatic hydrocarbon insoluble in water and soluble in many organic solvents.