What causes water to evaporate?
What causes water to evaporate?
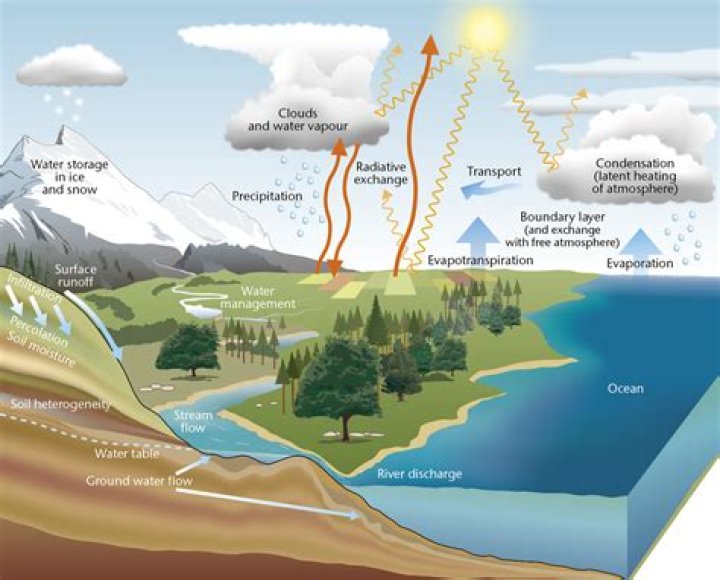
In the water cycle, evaporation occurs when sunlight warms the surface of the water. The heat from the sun makes the water molecules move faster and faster, until they move so fast they escape as a gas. When it is cool enough, the water vapor condenses and returns to liquid water.
Why does water evaporate or condense?
Evaporation and condensation happen when these molecules gain or lose energy. This energy exists in the form of heat. Evaporation happens when a liquid is heated. For example, as the sun heats water in a puddle, the puddle slowly shrinks.
Can water evaporate only?
Evaporation is a process of changing state from liquid to gaseous. Water is more common example , but all liquids can evaporate. E.g. alcohols, ketones, acids, etc.
Does water evaporate at night?
Yes there is evaporation at night. Water doesn’t have to be at 100°C to evaporate. The rate of evaporation may slow down when it’s cooler, but it will happen. All you need is the right combination of temperature, pressure and humidity.
At what temp does water evaporate?
212° F
Heat (energy) is necessary for evaporation to occur. Energy is used to break the bonds that hold water molecules together, which is why water easily evaporates at the boiling point (212° F, 100° C) but evaporates much more slowly at the freezing point.
Can humans evaporate?
The human body is a bit more complicated than a glass of water, but it still vaporizes like one. According to the captured study, it takes around three gigajoules of death-ray to entirely vaporize a person—enough to completely melt 5,000 pounds of steel or simulate a lightning bolt.
Does salt help evaporate water?
So, does saltwater evaporate? Yes, saltwater evaporates leaving behind salt crystals. And the addition of salt in pure water decreases the rate of evaporation of the salt solution.
What happens to water when it evaporates?
Evaporation happens when a liquid substance becomes a gas. When water is heated, it evaporates. The molecules move and vibrate so quickly that they escape into the atmosphere as molecules of water vapor. Once water evaporates, it also helps form clouds.
At what temperature does water stop evaporating?
100 deg C
The boiling point of water is 100 deg C at 1 atm. This is the temperature that liquid is in equilibrium with its gas phase. And you are right that below this temperature (at 1 atm) water should not vaporize.
Can water evaporate in dark?
Water don’t have any relation with light and evaporation. Water will evaporate even the dark room but if the lid is closed condensation will be seen on the top of the lid.
Why does the evaporation of water take place?
This explains why the water continues to evaporate. The point is that, when the vapour can be removed from the vessel containing the liquid, the normal equilibrium is not established and the water can slowly evaporate (at least as long as the surrounding environment has a lower humidity than the equilibrium vapour pressure).
What causes water to evaporate in a vacuum?
In a vacuum, this equilibrium will never happen, because the molecules in the vapor are immediately “diluted” away. This will actually cause the water not only to evaporate in a vacuum, but even to boil: bubbles of vapor will form inside the bulk of the liquid.
Why does water evaporate faster at the freezing point?
Heat (energy) is necessary for evaporation to occur. Energy is used to break the bonds that hold water molecules together, which is why water easily evaporates at the boiling point (212° F, 100° C) but evaporates much more slowly at the freezing point. Net evaporation occurs when the rate of evaporation exceeds the rate of condensation.
How does water evaporate if it doesn’t boil?
Steam is water in its gaseous phase. You can’t see water vapor, you can’t see steam, but you can see mist, which is liquid water droplets suspended in the air. When you boil water on the stove, you get steam. This then cools when it comes into contact with the air, increasing the relative humidity above 100%,…
Why is my water evaporating so fast?
Why does my pool water evaporate so fast? Evaporation occurs whenever you expose wind or air to the surface of your pool. Water molecules rise to the surface, form into a vapor and eventually get released into the air. Heated pools on cool nights experience evaporation more rapidly.
What causes the water to evaporate?
Causes of Evaporation. Evaporation occurs when liquid water turns into a water vapor, with about 90 percent of the water going through such a transformation originating from rivers, lakes and oceans. It’s easiest to understand the cause of evaporation by considering a pot of boiling water.
Why would water condense or evaporate?
Water on Earth’s surface will evaporate into the atmosphere as energy is absorbed by liquid water. Water molecules that exist in the liquid phase are free-flowing and in no particular fixed position. Once energy is added to water by heat from the sun, the bonds between the water molecules gain kinetic energy or energy in motion. They then escape the surface of the liquid and become a gas (water vapor), which then rises into the atmosphere.
What drives water evaporation?
Evaporation is an essential part of the water cycle. The sun (solar energy) drives evaporation of water from oceans, lakes, moisture in the soil, and other sources of water.

