What atom has 8 protons 9 neutrons and 10 electrons?
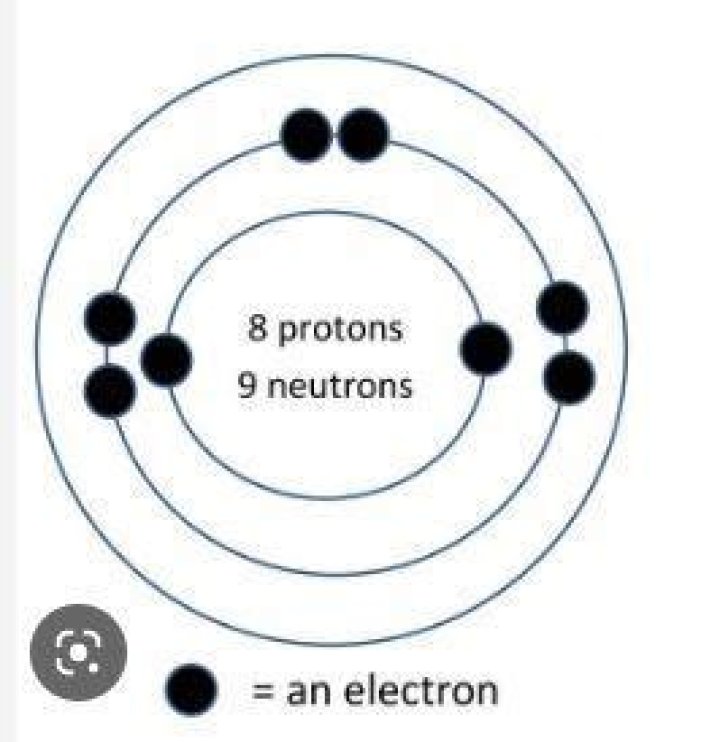
What atom has 8 protons 9 neutrons and 10 electrons?
O-16 has 8 protons and 8 neutrons O-17 has 8 protons and 9 neutrons O-18 has 8 protons and 10 neutrons. These are all isotopes of oxygen. Negatively charged particles “orbiting” the nucleus in discreet levels.
What atom has 8 protons 10 neutrons and 10 electrons?
18O2− has 8 protons, 10 neutrons, and 10 electrons.
What element has 8 protons 9 electrons?
Oxygen’s
Oxygen’s 8 electrons are negatively charged, and they orbit the atomic nucleus and balance the positive charge of the 8 protons. The positive charge of 1 proton exactly cancels the negative charge of 1 electron. Answer 9: Oxygen is atomic number 8 on the periodic table, which means it has 8 protons!
What symbol represents an atom with 8 protons and 10 electrons?
What symbol designates an ion that contains 8 protons and 10 electrons? O2- indicates an ion of oxygen having two extra electrons. I.e., since an oxygen atom normally has 8 protons and 8 electrons, this ion has 10 electrons (-10 charge) and 8 protons (+8 charge) giving it a charge of -2 (-10 + 8 = -2).
What element has 10 protons 10 electrons and 10 neutrons?
Neon
Neon is an atom with atomic number ten. Its atomic weight is 20.179 which cause it to have ten neutrons and ten protons in its nucleus and ten electrons outside.
What has 8 protons 9 neutrons and 8 electrons?
Oxygen has eight protons, eight neutrons, and eight electrons. Oxygen is different from carbon because it has more particles. Each kind of atom has a different number of the three particles. Protons and neutrons are in the center, or nucleus, of the atom.
What element has 9 protons 10 neutrons and 9 electrons?
Explanation: By looking at the periodic table, you will see that Fluorine has 9 protons. Since the number of electrons equal the number of protons, Fluorine has 9 electrons as well. Meanwhile, it’s mass number of 19, minus 10 neutrons, gives you 9 protons or electrons.
What is the symbol for an ion which has 9 protons and 10 electrons?
First, find F in the periodic table. Its atomic number, 9, indicates that there are 9 protons and 9 electrons in a neutral F atom. Use the equation E = P – C to calculate the number of electrons in an F- ion: E = 9 – (-1) = 10 electrons. So, an F – ion has 9 protons and 10 electrons.
What is the element with 8 protons?
Oxygen
Oxygen with the symbol O has the atomic number 8 which means it is the 8th element in the table. The number eight also means that oxygen has eight protons in the nucleus.
What is the total charge of 8 protons 8 neutrons and 10 electrons?
minus two
Explanation: With eight protons, we are necessarily looking at oxygen, but with 10 electrons, the net charge on the ion is minus two.
What element has 9 protons and 9 neutrons and 9 electrons?
element fluorine
The element fluorine has 9 protons and 9 electrons.
What element has 10 electrons 10 neutrons and 9 protons?
Since the number of electrons equal the number of protons, Fluorine has 9 electrons as well. Meanwhile, it’s mass number of 19, minus 10 neutrons, gives you 9 protons or electrons. Hence, the atom would be Fluorine.