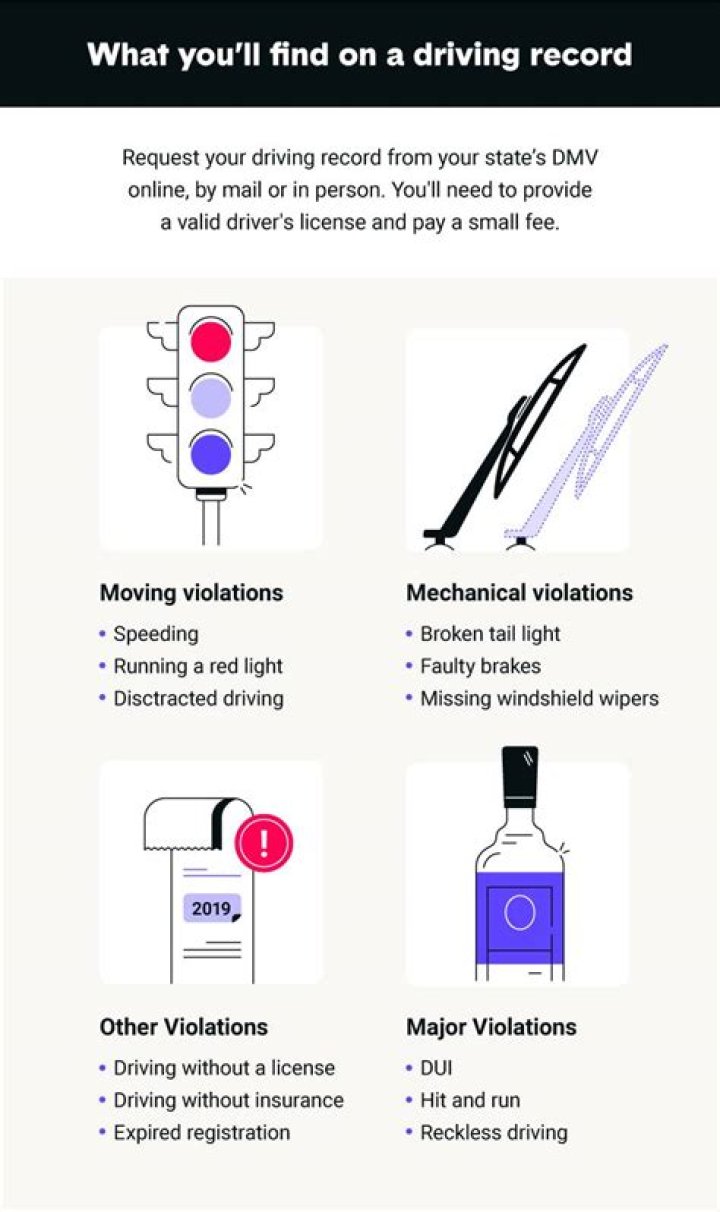
What are the rules for naming covalent molecules?
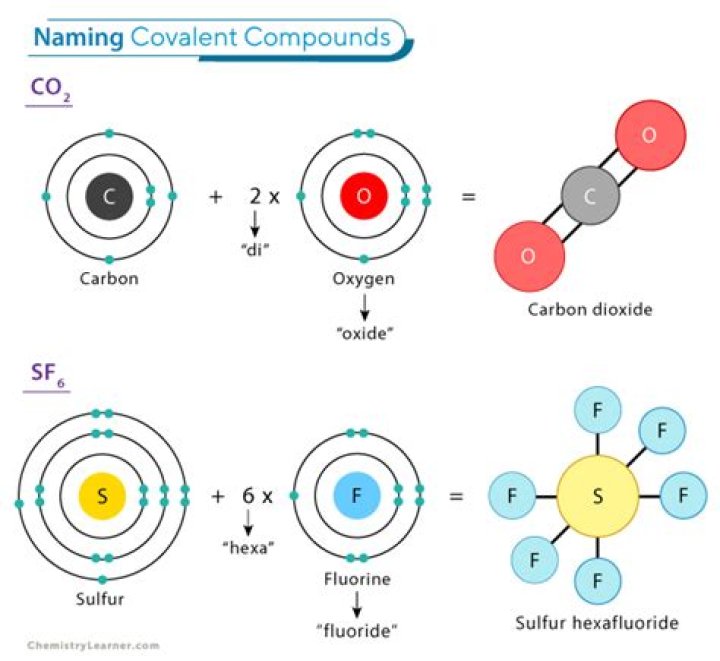
- Name the non-metal furthest to the left on the periodic table by its elemental name.
- Name the other non-metal by its elemental name and an -ide ending.
- Use the prefixes mono-, di-, tri-. to indicate the number of that element in the molecule.
.
Also know, what are the rules for naming and writing molecular compound formulas?
write the names of the elements in the order listed in the formula. Use prefixes to indicate the number of each atom. End the name of the second element with -ide. To write the formula of a binary molecular compound, use the prefixes to determine the subscript of each element.
Likewise, is co2 a covalent compound? Carbon dioxide is covalent bond. Carbon cannot form ionic bond because in the simplest sense carbon can neither lose 4 electrons for stablisation (Due to it's strong attraction to the nucleus) nor gain 4 electrons (Since it's small nucleus with 6 protons cannot manage extra 4 electrons).
Also asked, what is covalent naming?
Binary Inorganic Compounds Binary covalent compounds—covalent compounds that contain only two elements—are named using a procedure similar to that used for simple ionic compounds, but prefixes are added as needed to indicate the number of atoms of each kind.
Which element goes first in a covalent bond?
Answers. A covalent compound is usually composed of two or more nonmetal elements. It is just like an ionic compound except that the element further down and to the left on the periodic table is listed first and is named with the element name.
Related Question AnswersWhat is another name for a covalent compound?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.What are examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.
Are molecular and covalent compounds the same?
The terms covalent and molecular are often used interchangeably to describe the same type of substance. Covalent compounds are those that exhibit covalent bonding. Molecular compounds are a type of covalent compound. Molecular compounds exist as individual molecules.How do you make a covalent bond?
Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.What makes a molecular compound?
A molecular compound is usually composed of two or more nonmetal elements. Molecular compounds are named with the first element first and then the second element by using the stem of the element name plus the suffix -ide. Numerical prefixes are used to specify the number of atoms in a molecule.What is an example of a molecular compound?
Molecular Compounds. Molecular compounds are inorganic compounds that take the form of discrete molecules. Examples include such familiar substances as water (H2O) and carbon dioxide (CO2). In a carbon dioxide molecule, there are two of these bonds, each occurring between the carbon atom and one of the two oxygen atomsWhat are the steps in naming compounds?
4 Steps to Naming Compounds in Chemistry Nomenclature- Is it an Ionic Compound or a Molecular Compound? Ernest Wolfe.
- Add an 'ide' to the end of the second compound's name. For both molecular and ionic compounds, change the name of the second compound so it ends in 'ide'
- See if you need roman numerals.
- See if you need prefixes.
Why is oxygen a diatomic molecule?
Oxygen generally exists as a diatomic molecule in the atmosphere when it is not combined with any other element. It forms the molecule O2 because in that configuration, it has its lowest energy level when uncombined. All substances tend to go to the lowest energy level possible. Oxygen has 6 valence electrons.Do you use prefixes when naming ionic compounds?
Do not use numerical prefixes such as mono-, di-, tri-, etc. when naming ionic compounds — those are only used in naming covalent molecular compounds.Why is naming compounds important?
he primary function of chemical nomenclature is to ensure that a spoken or written chemical name leaves no ambiguity concerning which chemical compound the name refers to: each chemical name should refer to a single substance. The first separation of importance is to distinguish between inorganic and organic compounds.What are the prefixes for covalent bonds?
Here is a chart of those prefixes:- mono.
- di.
- tri.
- tetra.
- penta.
- hexa.
- hepta.
- octa.