What are alkali metals used in? Hydrogen is used in: hydrogen fuel, weather balloons and it creates water. Lithium is used in: Batteries, in the form of lithium carbonate it is used to control certain mental disorders and glass..
Similarly, it is asked, what are some uses of alkali metals?
Uses of alkali metals - definition
- Pure sodium has many applications, including use in sodium-vapor lamps which produce very efficient light .
- Potassium has a vital rolel in biological system. KCl used as a fertilizer while KOH is used in the manufacture of soap.
- Caesium is used making photoelectric cells.
Beside above, where are alkali metals found? Alkali metals are the chemical elements found in Group 1 of the periodic table. The alkali metals include: lithium, sodium, potassium, rubidium, cesium, and francium.
In respect to this, why are alkali metals so important?
Interesting Facts about Alkali Metals Because they are so reactive with air and water, they are generally stored in oil. Cesium and rubidium are used to make atomic clocks. Cesium clocks are considered the most accurate of all clocks. Sodium and potassium both play an important role in biological life on Earth.
What are the properties of alkali metals?
Characteristics of alkali metals are:
- High reactive metals.
- Not found freely in nature.
- Stored in a mineral oil solution.
- Low melting points.
- Low densities (lower than other metals)
- Low electronegativity.
- Low ionization energy.
- React easily with halogens.
Related Question Answers
What are common uses for alkaline earth metals?
Uses of Alkaline Earth Compounds Since magnesium burns brightly, it is used in flares and fireworks. Magnesium alloys with aluminum provide light weight and sturdy materials for airplanes, missiles, and rockets. Several antacids use magnesium hydroxide to neutralize excess stomach acid.How do you store alkali metals?
To minimize contact with oxygen and water, alkali metals must be stored in an airtight container under mineral oil and/or under an inert gas, such as argon. Glove boxes with an inert atmosphere are an appropriate location for the storage of alkali metals.Are alkali metals soft or hard?
The alkali metals are solids at room temperature (except for hydrogen), but have fairly low melting points: lithium melts at 181ºC, sodium at 98ºC, potassium at 63ºC, rubidium at 39ºC, and cesium at 28ºC. They are also relatively soft metals: sodium and potassium can be cut with a butter knife.Which element is the most reactive?
The most reactive metal on the periodic table is francium. Francium, however, is a laboratory-produced element and only minute quantities have been made, so for all practical purposes, the most reactive metal is cesium.Why are Group 2 called alkaline earth metals?
Group 1 elements are called alkali metals because their oxides are soluble in water and group 2 elements are called alkaline earth metals as their oxides form in the earth and are water soluble.Which alkali metal is most reactive with water?
Cesium
Which element has the highest ionization energy?
Fluorine
Why do alkali metals react to water?
The alkali metals are very reactive with water because they only have 1 electron in their outer shell when a stable outer shell contains 2 electrons. However, pure alkali metals will react with water to form a metal hydroxide and hydrogen gas. Colder water causes a more violent reaction.Why are they called alkaline earth metals?
The first term alkaline : They are called alkaline earth metals because they form alkaline solutions (hydroxides) when they react with water . The alkaline earth metals are named after their oxides, the alkaline earths, whose old-fashioned names were beryllia, magnesia, lime, strontia, and baryta.Is Lithium a metal?
Lithium. Lithium is part of the alkali metal group and can be found in the first column of the periodic table right below hydrogen. Like all alkali metals it has a single valence electron that it readily gives up to form a cation or compound. At room temperature lithium is a soft metal that is silvery-white in color.Why are halogens so reactive?
Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements.Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.Who discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.What are Group 2 elements called?
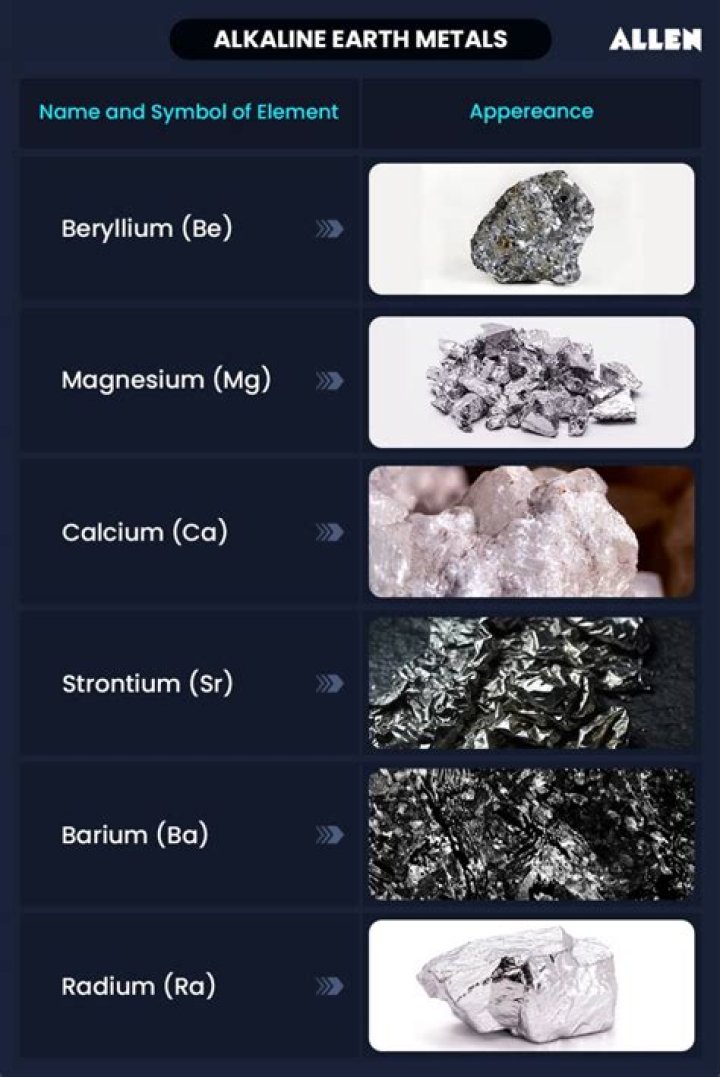
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).What do you mean by alkali metals?
Alkali metals are any of the elements found in Group IA of the periodic table (the first column). Alkali metals are very reactive chemical species that readily lose their one valence electron to form ionic compounds with nonmetals. All elements in the alkali metal group occur in nature.How are alkali metals used in society?
In its chemical form, it's so volatile that it can't be stored in anything other than kerosene. In the past, it has also been used, in the form of potash or potassium carbonate, to create soap and glass, and, in the form of potassium nitrate, to make gunpowder.What is unique about alkaline earth metals?
Interesting Facts about Alkaline Earth Metals They are called alkaline because they form solutions with a pH greater than 7, making them bases or "alkaline." Radium is formed from the decay of uranium. It is very radioactive and is dangerous to handle. Calcium and magnesium are important for animal and plant life.Are alkali metals rare?
The other alkali metals are considerably more rare, with rubidium, lithium, and cesium, respectively, forming 0.03, 0.007, and 0.0007 percent of Earth's crust. Francium, a natural radioactive isotope, is very rare and was not discovered until 1939. periodic tableModern version of the periodic table of the elements.How are alkali metals found?
Alkali metals are the first group in the periodic table. They are never found in nature uncombined because they are unstable and they react fast to other elements. They bond well with all elements except the noble gases. When they are in air, they quickly turn black.