What are examples of endergonic and exergonic reactions
Often, one chemical reaction feeds the next and endergonic reactions are coupled to exergonic reactions to give them enough energy to proceed. For example, firefly bioluminescence results from endergonic luminescence by luciferin, coupled with exergonic ATP release.
What is an example of an exergonic reaction?
Exergonic reactions occur spontaneously (no outside energy is required to start them). Examples of exergonic reactions include exothermic reactions, such as mixing sodium and chlorine to make table salt, combustion, and chemiluminescence (light is the energy that is released).
What are endergonic and exergonic reactions?
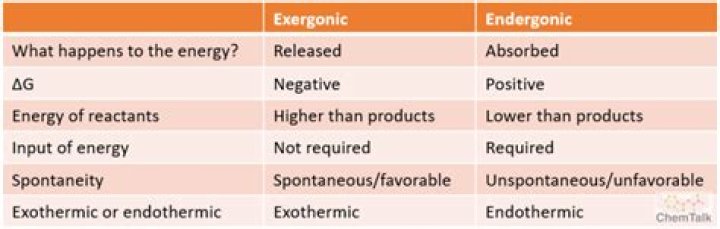
In an exergonic reaction, energy is released to the surroundings. The bonds being formed are stronger than the bonds being broken. In an endergonic reaction, energy is absorbed from the surroundings. The bonds being formed are weaker than the bonds being broken.
What is an example of endergonic?
One of the most common examples of an endergonic reaction is the process of photosynthesis. Photosynthesis is used by all plants to convert light energy into a form of chemical energy that can be used to fuel their life processes. Photosynthesis doesn’t happen spontaneously.What are some examples of endergonic reactions in a cell?
Examples of endergonic reactions in cells include protein synthesis, and the Na+/K+ pump which drives nerve conduction and muscle contraction.
Is photosynthesis an exergonic reaction?
Photosynthesis is an endergonic (energy-consuming) process. Cellular respiration is an exergonic (energy-releasing) process.
Is glycolysis endergonic or exergonic reaction?
The overall reaction of glycolysis is exergonic.
Is photosynthesis an endergonic?
Photosynthesis is an endergonic process. Photosynthesis takes in energy and uses it to build carbon compounds. … This diagram shows that photosynthesis is an endergonic reaction because it takes in energy.Are light reactions endergonic or exergonic?
The exergonic light-dependent reactions of photosynthesis convert light energy into chemical energy, producing ATP and NADPH. These reactions occur in the thylakoids of the chloroplasts. The products of the light-dependent reactions, ATP and NADPH, are both required for the endergonic light-independent reactions.
Is phosphorylation endergonic or exergonic?The phosphorylation (or condensation of phosphate groups onto AMP) is an endergonic process. By contrast, the hydrolysis of one or two phosphate groups from ATP, a process called dephosphorylation, is exergonic.
Article first time published onWhat are exergonic and endergonic reactions Slideshare?
Types of reactions • Exergonic is a spontaneous reaction that releases energy. If the free energy change is negative ,this reaction is due to loss of energy from reactants, so it is called exergonic. … If the free energy change is positive, the reaction is called endergonic.
Is sweating exergonic or endergonic?
When you sweat, the system – your body – cools down as perspiration evaporates from the skin and heat flows to the surrounding area. This means sweating is an exothermic reaction.
Is ATP endergonic or exergonic?
Not only is ATP hydrolysis an exergonic process with a large −∆G, but ATP is also a very unstable molecule that rapidly breaks down into ADP + Pi if not utilized quickly. This suggests a very low EA since it hydrolyzes so quickly.
Is osmosis endergonic or exergonic?
Active transport is the other class of transport systems that do require energy from the cell, usually in the form of ATP, do not occur spontaneously (endergonic reaction). Diffusion, osmosis, and facilitated diffusion are all types of passive transport.
What is an endergonic reaction in chemistry?
Endergonic: A process such as a chemical reaction in which the Gibbs free energy (G) of the products is greater than the Gibbs free energy of the reactants (i.e., ΔG > 0). Work (energy) must be absorbed from the surroundings for the reaction to proceed.
Is DNA replication endergonic or exergonic?
DNA replication is a exergonic process because of the hydrolysis of nucleotides. Monomers that serve as substrates in the reaction are deoxyribonucleoside triphosphates (dNTPs), which have a high potential energy due to their three closely packed phosphate groups, so DNA polymerization is exergonic.
Is gluconeogenesis endergonic or exergonic?
The gluconeogenesis pathway is highly endergonic until it is coupled to the hydrolysis of ATP or GTP, effectively making the process exergonic. For example, the pathway leading from pyruvate to glucose-6-phosphate requires 4 molecules of ATP and 2 molecules of GTP to proceed spontaneously.
Which reactions are endergonic in glycolysis?
The net result is the formation of two molecules of G-3-P in the last reactions of Stage 1 of glycolysis. The enzymes F-diP aldolase and triose-P-isomerase both catalyze freely reversible reactions. Also, both reactions proceed with a positive free energy change and are therefore endergonic.
Is Glycogenolysis exergonic or endergonic?
the breakdown of complex substances to simpler substances, occurs with a demand for energy and is exergonic (energy releasing).
What type of reaction is photosynthesis endergonic?
Photosynthesis is termed an endergonic reaction because it requires an input of energy in order to proceed.
Is photosynthesis an endergonic or exergonic reaction quizlet?
Is photosynthesis an endergonic or exergonic reaction? Explain why photosynthesis is an endergonic reaction. products have more energy than the reactants.
Is protein synthesis endergonic or Exergonic?
Protein synthesis is an endergonic process as it requires energy. Energy to drive the process is derived from the hydrolysis of ATP and GTP.
Are dark reactions Exergonic?
Their stored energy will be used to make sugar from carbon dioxide. These anabolic reactions are endergonic (have a positive delta G), and therefore require energy (from ATP and NADPH). … These reactions are sometimes called the “dark reactions” because they can occur in the dark (as long as ATP and NADPH are available).
Is photosynthesis is a combination reaction?
The photosynthesis chemical equation states that the reactants (carbon dioxide, water and sunlight), yield two products, glucose and oxygen gas. The single chemical equation represents the overall process of photosynthesis. …
Is photosynthesis catabolic Exergonic or anabolic Endergonic?
(c) What could be the raw material of this process? Out of respiration and photosynthesis which is anabolic and which is catabolic process.
Is photosynthesis an example of endergonic reaction?
An endergonic reaction is one that requires free energy to proceed. An example of an endergonic reaction of biological interest is photosynthesis. Photosynthetic organisms conduct this reaction by using solar photons to drive the reduction of carbon dioxide to glucose and the oxidation of water to oxygen.
Which type of reaction is photosynthesis?
Photosynthesis is an endothermic reaction. This means it cannot occur without energy (from the Sun). The light required is absorbed by a green pigment called chlorophyll in the leaves.
What's the reaction of photosynthesis?
The process of photosynthesis is commonly written as: 6CO2 + 6H2O → C6H12O6 + 6O2. This means that the reactants, six carbon dioxide molecules and six water molecules, are converted by light energy captured by chlorophyll (implied by the arrow) into a sugar molecule and six oxygen molecules, the products.
Is dehydration endergonic or exergonic?
A dehydration synthesis is an endergonic (or ‘energy in’) type of reaction that cannot take place without the input of energy from somewhere else. It is non-spontaneous, and by the second law of thermodynamics will not take place on its own.
Is hydrolysis exergonic or endergonic?
That hydrolysis is an exergonic reaction and it yields energy. The bonds holding the phosphate onto ATP are weak. They are known as high energy bonds but not because they are strong (if they were strong it would require alot of energy to break them.
Why is phosphorylation exergonic?
Explanation: Phosphorylation is the addition of a phosphate group to a molecule. This process requires energy because it results in new bonds being formed and a more complex product being created. Because the products are of a higher energy than the reactants, it is considered endergonic.