In chemistry, an ester is a chemical compound derived from an acid (organic or inorganic) in which at least one –OH (hydroxyl) group is replaced by an –O–alkyl (alkoxy) group. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones..
Similarly, you may ask, what are examples of esters?
Examples of Esters The hydrogen on the carboxyl group of acetic acid is replaced with an ethyl group. Other examples of esters include ethyl propanoate, propyl methanoate, propyl ethanoate, and methyl butanoate.
Secondly, how is an ester formed? Esters are formed by the condensation reaction between an alcohol and a carboxylic acid. This is known as esterification. In a condensation reaction, two molecules join and produce a larger molecule whilst eliminating a small molecule. During esterification this small molecule is water.
Herein, what are esters used for?
These and other volatile esters with characteristic odours are used in synthetic flavours, perfumes, and cosmetics. Certain volatile esters are used as solvents for lacquers, paints, and varnishes; for this purpose, large quantities of ethyl acetate and butyl acetate are commercially produced.
What is an ester and how is it formed?
Esters are formed when the carboxylic acid is heated with the alcohol in the presence of a catalyst. In this reaction, the concentrated sulphuric acid is used as a catalyst, dry form of hydrogen chloride gas is used in some cases. This method of reaction is used to convert alcohols into an ester.
Related Question Answers
Is COOH an acid or base?
Carboxyl groups are weak acids, dissociating partially to release hydrogen ions. The carboxyl group (symbolized as COOH) has both a carbonyl and a hydroxyl group attached to the same carbon atom, resulting in new properties.What are esters found in?
Esters are ubiquitous. Most naturally occurring fats and oils are the fatty acid esters of glycerol. Esters are typically fragrant, and those with low enough molecular weights to be volatile are commonly used as perfumes and are found in essential oils and pheromones.What products contain esters?
Esters in Food
| Ester | Flavor |
| N-amyl acetate | Pears, bananas |
| N-octyl acetate | Oranges |
| Methyl butrate | Apples |
| Ethyl butrate | Pineapples |
What are the main uses of esters?
There are various uses of esters. - Esters that are have fragrant odours are used as a constituent of perfumes, essential oils, food flavourings, cosmetics, etc.
- Esters are used as an organic solvent.
- Natural esters are found in pheromones.
- Naturally occurring fats and oils are fatty acid esters of glycerol.
How are esters named?
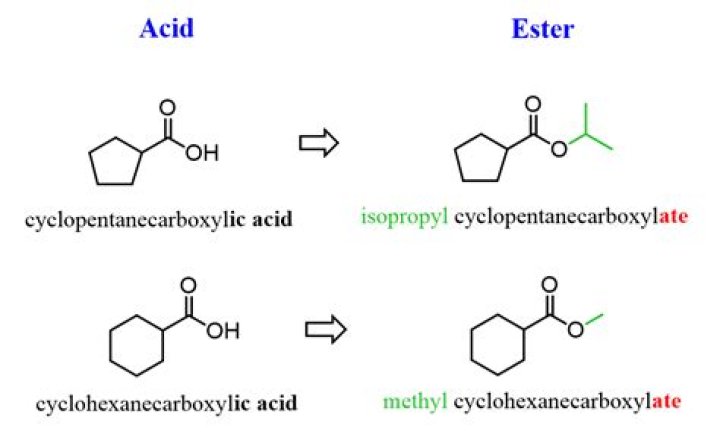
Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain from the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.Are esters toxic?
Inhalation of the fumes from some esters irritates the mucous membranes. Carboxylic esters have low to moderate toxicity via dermal and oral exposure. Some esters are used as flavoring agents in foodstuffs. The pyrophosphate esters (such as tetraethyl pyrophosphate) are highly toxic.How many esters are there?
Esters have the general formula RCOOR′, where R may be a hydrogen atom, an alkyl group, or an aryl group, and R′ may be an alkyl group or an aryl group but not a hydrogen atom. (If it were hydrogen atom, the compound would be a carboxylic acid.) Figure 15.5. 1 shows models for two common esters.Are esters acidic?
No, and esters are less acidic than water. Conclusion: esters are not acidic.Why are esters insoluble in water?
Esters can form hydrogen bonds through their oxygen atoms to the hydrogen atoms of water molecules. Thus, esters are slightly soluble in water. However, because esters do not have a hydrogen atom to form a hydrogen bond to an oxygen atom of water, they are less soluble than carboxylic acids.Who discovered esterification?
Emil Fischer
What is Ester value?
The ester value is the number of mg of potassium hydroxide required to saponify the esters in 1.0 g of the substance.Is Ester a functional group?
Esters are a functional group commonly encountered in organic chemistry. They are characterized by a carbon bound to three other atoms: a single bond to a carbon, a double bond to an oxygen, and a single bond to an oxygen. Esters react with nucleophiles at the carbonyl carbon.What is esterification process?
Esterification is a chemical reaction that forms at least one ester (= a type of compound produced by reaction between acids and alcohols). Esters are produced when acids are heated with alcohols in a process called esterification. An ester can be made by an esterification reaction of a carboxylic acid and an alcohol.Are esters used in ice cream?
Emulsifiers commonly used in ice cream include mono-diglycerides (E471), lactic acid esters (E472b), propylene glycol esters (E477) and blends of these.Why do esters smell?
Esters smell partly because they exhibit weak intermolecular forces. This allows ester molecules to enter the gas phase and reach your nose. These are no strongly positively polarized hydrogens in esters to participate in hydrogen bonding. Consider for example ethyl butyrate, which smells like pineapples.What are natural esters?
Natural esters are made from renewable natural sources, for example MIDEL eN 1204 (rapeseed/canola) and MIDEL eN 1215 (soybean). The base oil is chosen to give the best possible fit to the application; however unlike synthetic esters the properties of these base oils cannot be significantly altered.What type of reaction is esterification?
Esters and Formation of esters. Esters and water are formed when alcohols react with carboxylic acids. This reaction is called esterification, which is a reversible reaction. This type of reaction is called a condensation reaction, which means that water molecules are eliminated during the reaction.Why are esters not used in perfumes?
Esters often have a pleasant fruity aroma as can be seen in the chart to the right. However, that does not necessarily make them ideal for perfumes. Most simple esters give off these pleasant smells, but problems arise because they are not prepared to handle the sweat that a human body releases.How do you test for esters?
A simple way of detecting the smell of the ester is to pour the mixture into some water in a small beaker. Apart from the very small ones, esters are fairly insoluble in water and tend to form a thin layer on the surface. Excess acid and alcohol both dissolve and are tucked safely away under the ester layer.