What are common impurities in NMR?
What are common impurities in NMR?
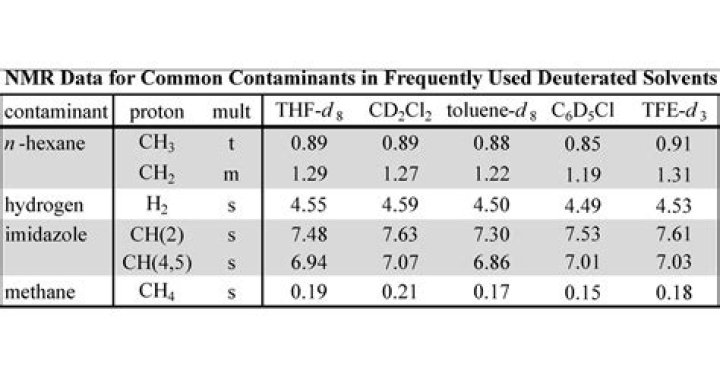
Building upon the work of Gottlieb, Kotlyar, and Nudelman in the Journal of Organic Chemistry, signals for common impurities are now reported in additional NMR solvents (tetrahydrofuran-d8, toluene-d8, dichloromethane-d2, chlorobenzene-d5, and 2,2,2-trifluoroethanol-d3) which are frequently used in organometallic …
Which solvent Cannot use NMR?
To avoid spectra dominated by the solvent signal, most 1H NMR spectra are recorded in a deuterated solvent….Notes on NMR Solvents.
| Solvent | 1H NMR Chemical Shift | 13C NMR Chemical Shift |
|---|---|---|
| Acetonitrile | 1.94 (5) | 118.7 (1) , 1.39 (7) |
| Benzene | 7.16 (1) | 128.4 (3) |
| Chloroform | 7.26 (1) | 77.2 (3) |
| Dimethyl Sulfoxide | 2.50 (5) | 39.5 (7) |
What is the chemical shift of water in CDCl3?
CDCl3 has ability to make hydrogen bond due to the polar bond between the C-Cl but not benzene. That is the reason water shows at 1.56 in cdcl3 and 0.40 in benzene.
Can NMR be used for purity?
As a primary analytical method, absolute quantitative 1H NMR spectroscopy offers certain advantages for purity determination. As samples and the standards are accurately weighed, inorganic impurities and other nonobservables will be detected and most solvent residues and water will be identified in the NMR spectrum.
How do you determine the purity of a substance?
Several physical properties can be used to identify a pure substance. Boiling point, melting point, color, density and solubility are examples of physical properties. One of the easiest ways to prove the purity of a substance is to compare the substance to a certified pure sample.
How does NMR choose solvent?
Factors to be considered when choosing a solvent are:
- Solubility: Clearly the more soluble the sample is in the solvent the better.
- Interference of solvent signals with the sample spectrum:
- Temperature dependence:
- Viscosity:
- Cost:
- Water content:
Which solvent is best for NMR?
How to Get a Good 1H NMR Spectrum
- Chloroform is the standard solvent to try first.
- If your compound is not soluble in chloroform, try benzene (nonpolar or average polarity compounds), acetone (dissolves almost anything) or methanol (polar compounds).
Why are deuterated solvents used in NMR spectroscopy?
Expensive deuterated solvents have traditionally been used for NMR spectroscopy in order to facilitate locking and shimming, as well as to suppress the large solvent signal that would otherwise occur in the proton NMR spectrum.
What is the chemical shift of the residual protons in deuterated chloroform on H NMR?
77.16 ppm
In carbon-13 NMR spectroscopy, the sole carbon in deuterated chloroform shows a triplet at a chemical shift of 77.16 ppm with the three peaks being about equal size, resulting from splitting by spin coupling to the attached spin-1 deuterium atom (CHCl3 has a chemical shift of 77.36 ppm).

