What are 5 physical properties of carbon?
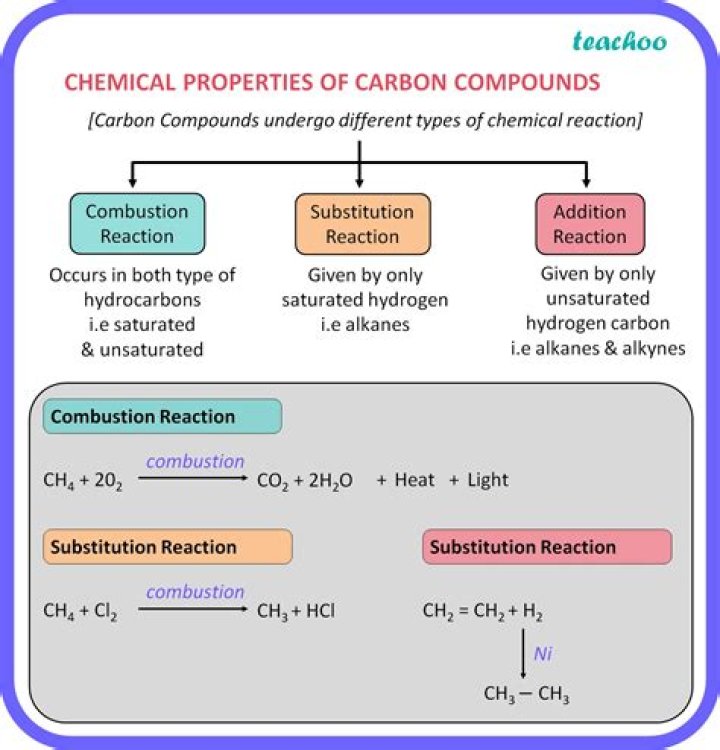
.
Keeping this in view, what are 4 properties of carbon?
Carbon has several allotropes, or different forms in which it can exist. These allotropes include graphite and diamond, which have very different properties. Despite carbon's ability to make 4 bonds and its presence in many compounds, it is highly unreactive under normal conditions.
Secondly, what are some physical and chemical properties of carbon dioxide? Carbon dioxide is a colorless & odorless gas. It is soluble in water, ethanol , acetone and has the following properties : Melting Point : -55.6 degC. Boiling Point : -78.5 degC.
Also to know is, what are 5 characteristics of carbon?
Carbon's characteristics include its ability to bond with oxygen, hydrogen, nitrogen, phosphorus and sulfur. Carbon biochemical compounds are essential to all life on the planet. Because of its bonding ability, carbon can form single, double, or triple covalent bonds with other atoms.
What are the characteristics of carbon?
Characteristics: Carbon can exist with several different 3 dimensional structures in which its atoms are arranged differently (allotropes). Three common crystalline allotropes are graphite, diamond, and (usually) fullerenes.
Related Question AnswersIs carbon a good conductor?
The form of carbon that is a good conductor of electricity is Graphite. Graphite is an allotrope of carbon that has the properties of both metal and non metal. The reason why graphite is a good conductor of electricity is because of the mobility of the electrons in its outer valence shell.Is carbon bad for your health?
Health effects of carbon Elemental carbon is of very low toxicity. Health hazard data presented here is based on exposures to carbon black, not elemental carbon. Chronic inhalation exposure to carbon black may result in temporary or permanent damage to lungs and heart.When was carbon first discovered?
Antoine LavoisierWhat is carbon made of?
What is carbon? Carbon is a chemical element, like hydrogen, oxygen, lead or any of the others in the periodic table. Carbon is a very abundant element. It exists in pure or nearly pure forms – such as diamonds and graphite – but can also combine with other elements to form molecules.Why is co poisonous?
Carbon monoxide is harmful when breathed because it displaces oxygen in the blood and deprives the heart, brain and other vital organs of oxygen. Large amounts of CO can overcome you in minutes without warning — causing you to lose consciousness and suffocate.Is coal a carbon?
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements; chiefly hydrogen, sulfur, oxygen, and nitrogen. The largest consumer and importer of coal is China.How is carbon created?
All the carbon atoms in the human body were created in the stars. Elementary particles, such as protons, were formed during the "big bang"; that amazing moment about 14 billion years ago in which the universe got it's start. Their creation had to come later in a dying star.What does carbon smell like?
Carbon dioxide is a colorless gas that is heavier than air. At low concentrations, carbon dioxide gas has no odor. At high concentrations, it has a sharp, acidic smell. At normal temperatures, carbon dioxide Welders use carbon dioxide to help prevent impurities, like rust, from forming at welds.What percentage of the universe is carbon?
Heavier elements were mostly produced much later, inside of stars. Hydrogen and helium are estimated to make up roughly 74% and 24% of all baryonic matter in the universe respectively.Universe.
| Z | Element | Mass fraction (ppm) |
|---|---|---|
| 6 | Carbon | 4,600 |
| 10 | Neon | 1,340 |
| 26 | Iron | 1,090 |
| 7 | Nitrogen | 960 |

