Is Protease the same as pepsin?
.
Simply so, is protease and pepsin the same thing?
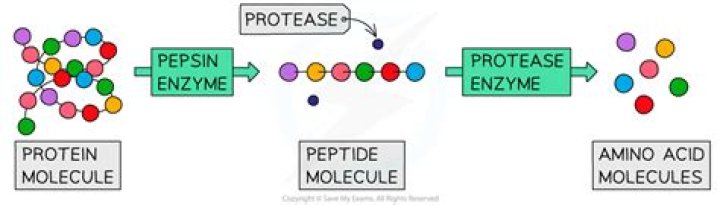
Pepsin is a type of protease which serves as the main digestive enzyme in the stomach. On the other hand, proteases are the protein digestive enzymes which break down proteins into small peptides and amino acids.
Also, what is the difference between protease and proteinase? Proteases (also called peptidases) are enzymes that degrade proteins by hydrolysis of peptide bonds. Endopeptidases break bonds of nonterminal amino acids (within the protein) while exopeptidases break bonds of terminal amino acids (the end of the protein). Proteinase is synonymous with endopeptidase.
Simply so, what type of protease is pepsin?
Pepsin is an aspartic protease, using a catalytic aspartate in its active site. It is one of three principal proteases in the human digestive system, the other two being chymotrypsin and trypsin.
Does Pepsin work in a basic environment?
pH and Enzyme Function. For example, the enzyme pepsin, which works in your stomach, functions best in a strongly acidic environment. Lipase, an enzyme found in your small intestine, works best in a basic environment. When the pH changes, the active site progressively distorts and affects enzyme function.
Related Question AnswersWhat foods contain pepsin?
Foods that contain natural digestive enzymes include pineapples, papayas, mangoes, honey, bananas, avocados, kefir, sauerkraut, kimchi, miso, kiwifruit and ginger. Adding any of these foods to your diet may help promote digestion and better gut health.What happens if pepsin is not present?
Answer and Explanation: Pepsin denatures ingested protein and converts it into amino acids. Without pepsin, our body would be unable to digest proteins.What is a protease enzyme?
Proteolytic enzyme, also called protease, proteinase, or peptidase, any of a group of enzymes that break the long chainlike molecules of proteins into shorter fragments (peptides) and eventually into their components, amino acids.What is the difference between pepsin and trypsin?
The main difference between pepsin and trypsin is that the pepsin is secreted by the gastric glands of the stomach whereas the trypsin is secreted by the exocrine glands of the pancreas. Pepsin and trypsin are two types of proteolytic enzymes secreted by the digestive system in order to digest proteins.What is pepsin good for?
Pepsin, the proteolytic enzyme of the stomach is normally responsible for less than 20% of the protein digestion occuring the gastrointestinal tract. It is an endopeptidase enzyme that metabolizes proteins to peptides. It preferentially hydrolyzes peptide linkages where one of the amino acids is aromatic.Is pepsin active in the mouth?
Would pepsin be active in the mouth? Explain your answer. No, since pH of mouth is closer to neutrality, you would expect pepsin to be slightly active, but not AS active as in the stomach with a pH of 2.Is pepsin made from pork?
The description states that the pepsin is from a fungal source with activity equivalent to animal derived pepsin. The Pepsin Enzyme & Gelatin would both be animal sourced, including pig.Why is pepsin most effective in the stomach?
The reason pepsin functions best at pH 2 is because the carboxylic acid group on the amino acid in the enzyme's active site must be in its protonated state, meaning bound to a hydrogen atom. At low pH the carboxylic acid group is protonated, which allows it to catalyze the chemical reaction of breaking chemical bonds.What is an example of protease?
Proteases are a protein-digestive enzyme that cleaves protein through hydrolysis, the addition of water to the peptide bond. An example of a protein-digesting enzyme may be seen in the protease called pepsin. Pepsin is one of two components of gastric juice. Pepsin works by attacking the exposed peptide bonds.How do you neutralize pepsin?
Also avoiding carbonated beverages, tomato-based products, citrus products, spicy foods, chocolate, breath mints, coffee, caffeinated beverages and alcohol reduces the activation of pepsin. I recommend drinking alkaline water with a pH greater than 9.5 to reduce the activation of the pepsin enzyme in the stomach.Can pepsin cause reflux?
Although acid is indeed one of the culprits in cases of gastroesophageal reflux disease (GERD), a growing body of evidence suggests that pepsin, an enzyme, is the substance that causes the most damage when the reflux extends beyond the upper esophagus and reaches the pharynx, larynx and lungs.How does pH affect pepsin?
The activated enzyme then acts autocatalytically to increase the rate of formation of more pepsin. It provides the appropriate pH for the enzyme to act. The optimum pH for pepsin is approximately pH 3.5. It denatures ingested protein; denatured protein is a better substrate for the enzyme than native protein.How is pepsin produced?
Pepsin is the chief digestive enzyme in the stomach that breaks down proteins. We see that chief cells produce pepsinogen (an inactive form of pepsin). Pepsinogen is converted to pepsin when the parietal cells found within the gastric glands secrete hydrochloric acid.Where is pepsin active in the body?
Pepsin Pearls Pepsin is an enzyme present in the stomach that digests proteins found in ingested food. A low pH (1.5 to 2) activates pepsin. Parietal cells within the stomach lining secrete hydrochloric acid that lower the pH of the stomach and activate pepsin.Is pepsin a peptidase?
Pepsin is a type of peptidase (or protease).What is the substrate for pepsin?
Enzyme - General Information| Enzyme | Substrate | Products |
|---|---|---|
| Lipase | Lipid | Glycerol + Fatty Acid |
| Enzyme | Substrate | Products |
| Pepsin | Protein | Peptides + Amino Acids |
| Trypsin | Protein | Peptides + Amino Acids |

