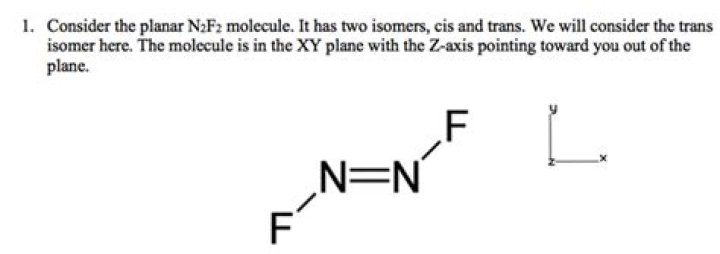
One way to solve this problem is to treat both of the nitrogen atoms as central atoms. However, because two of these pairs are in a double bond each nitrogen atom could be treated as having 3 electron pairs. This leaves two possible configurations: trigonal pyramidal and trigonal planar..
Similarly, is n2f2 polar?
In trans- , each bond is polar and the dipole moment of two bonds cancels out the dipole moment due to two lone pairs of electrons on each nitrogen atom. Thus, trans - is non polar molecule. In both the isomers, the nitrogen atom is in a non-linear environment with one lone pair of electrons.
Also Know, what is the name of n2f2? Dinitrogen difluoride is a chemical compound with the formula N2F2. It has the structure F−N=N−F and exists in both a cis- and trans-form.
Secondly, what shape is n2f2?
As we saw trigonal planar arrangements in N2F2, the nitrogen atoms are sp2-hybridized! The fluorine atoms are sp3-hybridized, since they carry three lone pairs of electrons each, and are bound to one atom only (either of the nitrogen atoms).
Does n2f2 follow the octet rule?
N has five valence electrons so it can form three bonds to satisfy its octet rule. In N2F2, one N is bonded to one surrounding F sharing one electron, so to satisfy its octet rule N has two electrons which are bonded to neighbour N directly with its two electrons, that's why there is double bond between N=N in N2F2.
Related Question Answers
Does becl2 have net dipole moment?
Step 5: Determine the polarity of the molecule. Step 1: Beryllium (EN = 1.5) is less electronegative than chlorine (EN = 3) so beryllium is the central atom. This means there is no net dipole in the molecule, thus BeCl2 has no dipole moment.What does Vsepr stand for?
Valence Shell Electron Pair Repulsion
What is the correct molecular formula for the compound dinitrogen dichloride?
N2O5
Which elements are exceptions to the octet rule?
The two elements that most commonly fail to complete an octet are boron and aluminum; they both readily form compounds in which they have six valence electrons, rather than the usual eight predicted by the octet rule.What are some exceptions to the octet rule?
However, there are three general exceptions to the octet rule: Molecules, such as NO, with an odd number of electrons; Molecules in which one or more atoms possess more than eight electrons, such as SF 6; and. Molecules such as BCl 3, in which one or more atoms possess less than eight electrons.What is an example of the octet rule?
Covalent Example For the carbon dioxide molecule, for example, one carbon atom bonds with two oxygen atoms. Carbon, with four electrons in its valence shell, needs four to reach the octet state. Oxygen, with six outer electrons, needs two.When can you break the octet rule?
However, there are three general exceptions to the octet rule: (1) molecules, such as NO, with an odd number of electrons; (2) molecules in which one or more atoms possess more than eight electrons, such as SF 6; and (3) molecules such as BCl 3, in which one or more atoms possess less than eight electrons.Does BeCl2 follow the octet rule?
BeCl2 violate octet rule. The boron must be in a suitable valence state to bind to the three chlorines. In the molecule the boron is associated with only six electrons.How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.Why is the octet rule not universal?
The octet rule is a rule of thumb from a very simple, qualitative bonding model that has wide utility - Lewis structures. But the rule is not universal, because it's limited to situations in which an atom is most stable with 8 valence electrons.Do resonance structures have to follow the octet rule?
All resonance structures must have the same number of valence electrons. The rule is violated above because structure E has 12 valence electrons and structure F has 14 valence electrons. So E and F are not resonance structures (F also violates the octet rule).Which compound does not follow octet rule?
Hydrogen and Lithium However, many atoms below atomic number 20 often form compounds that do not follow the octet rule. For example, with the duet rule of the first principal energy level, the noble gas helium, He, has two electrons in its outer level.