How many isomers of pentane are possible?
By Sophia Aguilar •
three
.
Similarly one may ask, how many isomers are in pentane?
three isomers
Subsequently, question is, how many isomers are there for c5h12? three
Just so, what are the possible isomers of pentane?
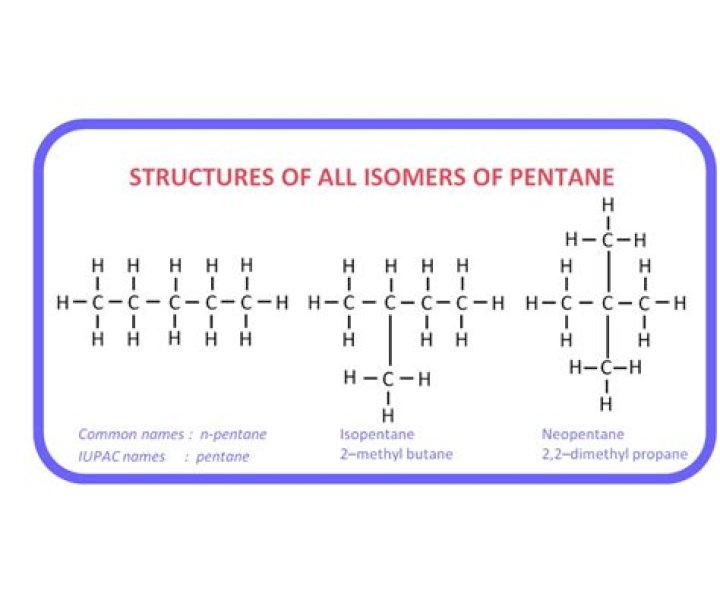
There are 3 known isomers of pentane:
- n-pentane - straight chain of 5 carbon atoms.
- methybutane (isopentane) - one methyl group attached to the 2ndcarbon of the main chain of four.
- dimethylpropane (neopentane) - 2 methyl groups thatched to thecentral carbon of a 3 carbon central chain (tetrahedral inshape).
What does C pentane smell like?
Pentane. Under normal conditions, Pentaneis a colourless liquid, which has a characteristicsmell. It evaporates easily at room temperature and boils at36 degrees celsius. Pentane is highly flammable and mixturesof the vapour with air can be explosive.
Related Question AnswersIs pentane a polar molecule?
Brother see this For example, this guideline could beused to predict that ethanol, which is composed of polarmolecules, would be soluble in water, which is also composed ofpolar molecules. Likewise, pentane (C5H12), which hasnonpolar molecules, is miscible with hexane, which also hasnonpolar molecules.Is pentane a hydrocarbon?
Isopentane is more stable than pentane by 1.8kcal/mol, and neopentane by 5 kcal/mol. A related cycloalkane iscyclopentane, although this is not an isomer of pentane asits molecular formula is different, cyclopentane is an isomer ofthe pentenes. Being an unfunctionalized hydrocarbon,pentane is a commodity chemical.What is n Pentane used for?
The properties of pentane are very similar tobutane and hexane. As well as being used as fuel,pentane has numerous industrial uses. Primarily,pentane is used to create a blowing agent which isthen used to create a foam known aspolystyrene.How many structural isomers are there?
Structural Or ConstitutionalIsomers With the structural formulaC4H10 there are two different isomerspossible.Is pentane an alkene?
A two-carbon chain is called ethane; a three-carbonchain, propane; and a four-carbon chain, butane. Longer chains arenamed as follows: pentane (five-carbon chain), hexane (6),heptane (7), octane (8), nonane (9), and decane (10).What c6h14 called?
2-Methylpentane is a branched-chain alkane with themolecular formula C6H14. It is a structural isomer of hexanecomposed of a methyl group bonded to the second carbon atom in apentane chain.Does pentane dissolve in water?
Is pentane soluble in water? Why or why not? Theshort answer is no. The longer answer is that you can onlydissolve about 40 mg of pentane in one Liter ofwater at 20 °C.Which pair of compounds are isomers?
Ch 1: Constitutional Isomers. Compoundsthat have the same molecular formula but different chemicalstructures are called isomers. Remember isomerism isa property between a pair (or more) of molecules, i.e. amolecule is an isomer of another molecule.Are the structural isomers possible for Bromopentane?
Yes, isomer of bromopentane ispossible. Structure of one of the isomers ofbromopentane is given above.What are the structural isomers of c5h12?
Hence the possible isomers are:- n-Pentane (Maximizing the number of carbon atoms in the mainchain)
- Isopentane or 2-methylbutane (Shortening the main chain to fourC-atoms and looking for positions to place one methyl group)
- Neopentane or 2,2-dimethylpropane (Main chain has three carbonsin this case)
Why boiling point of n Pentane is higher than isopentane?
The n-pentane melts higher thanisopentane because it can assume a rodlike shape in whichadjacent molecules can pack closely together. As a general rule,the more symmetrical the molecule, the higher itsmelting point and the lower its boiling pointrelative to its isomers.How many isomers of hexane are possible?
fiveWhere does Pentane come from?
Pentane is derived from the petroleum rawmaterials, natural gas and crude oil. It is generally marketed in acompressed liquefied form at 99% purity. n-Pentane is aflammable liquid.What is pentane plus?
Pentane plus is a natural gas liquid.Pentane is a key raw material followed by heptane, benzene,hexane and octane. Apart from natural heavy crude, pentaneplus is also as an additive for other oil mixes for smooth andeasy pipeline transportation.What is the condensed structural formula for pentane?
In IUPAC nomenclature however the term pentane isused exclusively for n-pentane, the unbranched isomer of thegroup. The molecular formula of pentane, orn-pentane, is C5H12 . The condensed structuralformula will be CH2(CH2)3CH3 .What are the 3 types of isomers?
There are two main types of isomers, structural isomers andstereoisomers (illustrated below).- Structural isomers.
- Stereoisomers.
- Methoxyethane and Propanol.
- Methylacetylene and Allene.
- Fulminate and Cyanate.
- Glucose and Fructose.
- Pharmaceutical Examples.
- Enzymatic Reactions.

