How is the percent composition of a compound calculated?
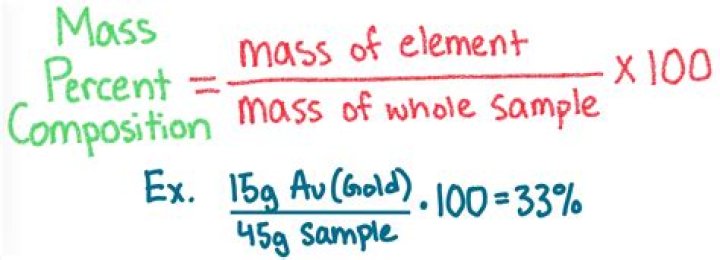
How is the percent composition of a compound calculated?
Therefore, percent composition of a compound is the percent by mass of each element in the total mass of a compound. Another way of saying the same thing is how much in percent each element contributes to the total mass of a compound. The higher the percentage composition, the higher the mass of the element present in the compound.
How to find the composition of an element?
If you do, you will get: 100% -11.21% = 88.79%. Once you know the percentage composition of all the elements in a chemical formula, except one. You can always use this shortcut to find its percentage composition.
When to use percent composition and density in chemistry?
Determining Percent Composition requires knowing the mass of entire object or molecule and the mass of its components. In the laboratory, density can be used to identify an element, while percent composition is used to determine the amount, by mass, of each element present in a chemical compound.
How to calculate the percentage of iron in a compound?
Step one – Work out the M r of the compound. Step three – Divide the total A r of the element by the M r of the compound, then multiply by 100. Find the percentage of iron in iron (III) oxide, Fe2O3. Step one – Work out the M r of the compound. Step three – Divide the total A r of the element by the M r of the compound, then multiply by 100.
How do you calculate percent composition?
Percent Composition by Mass. Percent composition is calculated from a molecular formula by dividing the mass of a single element in one mole of a compound by the mass of one mole of the entire compound. This value is presented as a percentage.
How do you calculate percent composition in chemistry?
The percent composition of a compound is calculated with the molecular formula: divide the mass of each element found in one mole of the compound by the total molar mass of the compound. The percent composition of a compound can be measured experimentally, and these values can be used to determine the empirical formula of a compound.
What is the formula for percent composition?
The formula for percent composition is: % by mass = (mass of element in the compound/ total mass of the compound) X 100.
What is an example of percent composition?
Updated August 11, 2019. Percent composition is the percentage by mass of each element in a compound. Example: The percent composition of water is 20% hydrogen and 80% oxygen.

