How is copper carbonate formed?
By Isabella Ramos •
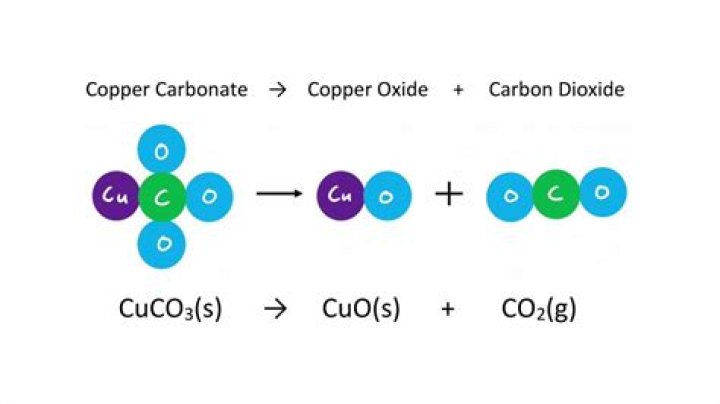
Reactions. Basic copper carbonate is decomposed by acids, such as solutions of hydrochloric acid HCl, into the copper(II) salt and carbon dioxide. In 1794 the French chemist Joseph Louis Proust (1754–1826) thermally decomposed copper carbonate to CO2 and CuO, cupric oxide.
.
Also to know is, how is CuCO3 formed?
CuCO3 ] is a chemical compound formed when a piece of copper left exposed in the atmosphere thus resulting in the corrosion of copper. This happens because copper reacts with water and carbon dioxide present in air.
Furthermore, does copper carbonate dissolve water? Acetic acid
what is copper carbonate used for?
Uses. Copper(II) carbonate can be used to kill algae. It is also used as a pigment.
Is copper carbonate a base?
Copper Carbonate is a very weak base. CuCO3 does not exist. What is commonly called copper carbonate is Cu(OH)2. And yes, that is basic.
Related Question AnswersIs CuCO3 an acid or base?
Calcium carbonate, being the salt of a strong base and weak acid, is considered a base. It reacts with avid to give out Carbon dioxide and water. Calcium carbonate (CaCO3) is an insoluble salt which means it dissolves only slightly in water.Is copper carbonate insoluble in water?
Copper carbonate is an insoluble solid. It is hard to recover the copper since the carbonate is unable to dissolve into copper ions and carbonate ions. When an acid is added to a carbonate water, carbon dioxide and a salt are formed.Is caco3 soluble in water?
Calcium carbonate has a very low solubility in pure water (15 mg/L at 25°C), but in rainwater saturated with carbon dioxide, its solubility increases due to the formation of more soluble calcium bicarbonate. Calcium carbonate is unusual in that its solubility increases as the temperature of the water decreases.Is copper carbonate a precipitate?
Description: Copper carbonate precipitate. When 0.25M copper sulfate (CuSO4) is poured into a beaker with 0.25M sodium carbonate (Na2CO3), blue precipitate of copper carbonate (CuCO3) is formed. The reaction is CuSO4 + Na2CO3 -> CuCO3 + Na2SO4.What is the color of copper carbonate?
Copper(II) carbonate is a powdery blue-green compound that is insoluble in water. Its color can be different shades of blue or green depending on the purity and the presence of other basic copper carbonates, which are usually present in any technical grade samples.Why is CuCO3 insoluble in water?
Is CuCO3 (Copper (II) carbonate) soluble or insoluble in water? The answer is that it is insoluble in water. Although it is an ionic compound, it does not dissociate into its ions in water. Zinc carbonate is therefore insoluble in water.What happens if you eat copper carbonate?
Harmful if swallowed. Ingestion may cause gastrointestinal irritation, nausea, vomiting and diarrhea. Ingestion of product may result in metal fume fever, a flu - like illness. Symptoms of metal fume fever may include fever, fatigue, vomiting, muscle aches and shortness of breath.How does copper turn green?
Copper turns green because of chemical reactions with the elements. Just as iron that is left unprotected in open air will corrode and form a flaky orange-red outer layer, copper that is exposed to the elements undergoes a series of chemical reactions that give the shiny metal a pale green outer layer called a patina.What does copper carbonate contain?
Copper(II) carbonate, also known as cupric carbonate, is a chemical compound. Its chemical formula is CuCO3. It contains copper in its +2 oxidation state. It also contains carbonate ions.How is basic copper carbonate formed?
Reactions. Basic copper carbonate is decomposed by acids, such as solutions of hydrochloric acid HCl, into the copper(II) salt and carbon dioxide. In 1794 the French chemist Joseph Louis Proust (1754–1826) thermally decomposed copper carbonate to CO2 and CuO, cupric oxide.What is the formula for basic copper carbonate?
Cu2(OH)2CO3Where is copper carbonate found?
Both malachite and azurite can be found in the verdigris patina that is found on weathered brass, bronze, and copper.Basic copper carbonate.
| Names | |
|---|---|
| Appearance | green powder |
| Density | 4 g/cm3 |
| Melting point | 200 °C (392 °F; 473 K) |
| Boiling point | 290 °C (554 °F; 563 K) decomposes |

