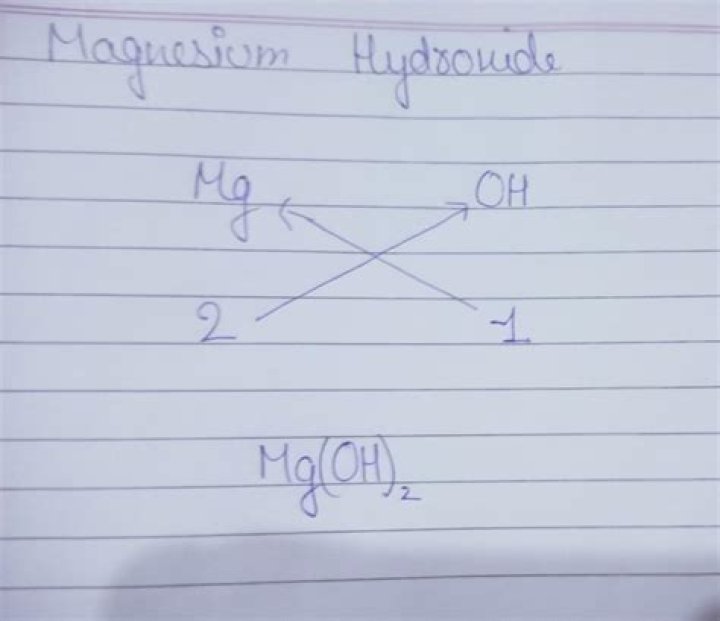
Magnesium hydroxide is a non-toxic inorganic base. Its aqueous suspension is known as milk of magnesia, a common antacid. Formula and structure: The chemical formula of magnesium hydroxide is Mg(OH)2, and its molar mass is 58.32 g/mol..
People also ask, what is the formula of magnesium?
Magnesium ion | Mg+2 - PubChem.
Furthermore, what two solutions could you use to make magnesium hydroxide? Magnesium chloride in the sea water is reacted with lime (calcium oxide) to form the Mg2+ and OH- ions, which precipitate into magnesium hydroxide salt. Another method is the electrolysis of fused magnesium chloride, which also gives Mg(OH)2 in the same way.
One may also ask, how is magnesium hydroxide made?
structure and properties Magnesium hydroxide, Mg(OH)2, is a white powder produced in large quantities from seawater by the addition of milk of lime (calcium hydroxide). It is the primary raw material in the production of magnesium metal and has been used as a fire-retardant additive.
How do you take magnesium hydroxide?
Take the tablets and capsules with a full glass of water. Chew the chewable tablets thoroughly before swallowing them. Shake the suspension well before measuring a dose. To ensure that you get the correct dose, measure the liquid form of magnesium with a dose-measuring spoon or cup, not with a regular table spoon.
Related Question Answers
What is the formula for metal?
They include such familiar metals as copper (Cu), silver (Ag), gold (Au) and iron (Fe). When you see the name of one of these metals in the name of a chemical formula, you'll probably also see the number in Roman numerals written after it to tell you the ionic charge the metal displays in the compound.What is magnesium used for?
Water with a high mineral content, or "hard" water, is also a source of magnesium. Magnesium is most commonly used for constipation, as an antacid for heartburn, for low magnesium levels, for pregnancy complications called pre-eclampsia and eclampsia, and for a certain type of irregular heartbeat (torsades de pointes).What does mg2+ mean?
Mg2+ is a Mg atom that now has the same number of electrons as a noble gas. It achiveved this by giving up to other atoms in its vicinity who could be considered “electron acceptors”: they are atoms from chemical elements that are ready to accept these electrons to reach the configuration of a noble gas.What is the color of magnesium?
In standard conditions magnesium is a light metal with a silvery-white color. When exposed to air, magnesium will tarnish and become protected by a thin layer of oxide. When coming into contact with water, magnesium will react and produce hydrogen gas. If submerged in water, you will see gas bubbles start to form.What is the chemical name for magnesium?
Magnesium. Chemical element, metallic, symbol Mg, situated in group IIa in the periodic table, atomic number: 12, atomic weight: 24,312. Magnesium is silvery white and very light.Why does magnesium have a 2+ charge?
Because magnesium is one of the group llA (Alkaline earth metal) element of the periodic table. As magnesium has lost 2 electrons so the electrons will be lesser than the number of protons present in nucleus by 2 number so it will no more be remained Neutral and will show 2 electrons scarce by +2 charge.Where can magnesium be found?
Biological metal On Earth, magnesium is found both in the crust and in the mantle; it's also the third-most abundant mineral dissolved in seawater, with a 0.13 percent concentration.What is the Valency of magnesium?
Answer and Explanation: The valency of magnesium is 2. Magnesium has an atomic number of 12 and it belongs to the second group in the periodic table.Is magnesium hydroxide safe on skin?
More safety Information: Concentrated Sodium Hydroxide, Calcium Hydroxide, Magnesium Hydroxide and Potassium Hydroxide are strong irritants and corrosive to the skin, eyes, respiratory tract and gastrointestinal system if ingested.Why is milk of magnesia banned in Australia?
Apparently it is banned in Australia – perhaps because chronic (constipated) users with renal dysfunction are unable to clear the excess Mg from the system. Although Magnesium is a minority element in the body (about 25g each per adult), it is crucial for the regulation of calcium and potassium flux.What is the pH value of magnesium hydroxide?
approximately 10
Is magnesium hydroxide healthy?
Magnesium hydroxide reduces stomach acid, and increases water in the intestines which may induce bowel movements. Magnesium hydroxide is used as a laxative to relieve occasional constipation. Magnesium hydroxide is also used as an antacid to relieve indigestion, sour stomach, and heartburn.Is magnesium hydroxide harmful?
Toxicity Profile for Magnesium hydroxide (1993) In man, magnesium hydroxide can cause eye damage. It is of low acute oral toxicity in humans. The principal effect at high doses is diarrhoea, although other symptoms may occur in subjects with poor kidney function, in whom urinary excretion of magnesium is impaired.Is magnesium hydroxide strong or weak base?
The solid mineral form of magnesium hydroxide is known as brucite. Since the dissociation of this small amount of dissolved magnesium hydroxide is complete, magnesium hydroxide is considered a strong electrolyte. Its low solubility makes it a weak base.What is the common name of magnesium hydroxide?
Magnesium hydroxide is the inorganic compound with the chemical formula Mg(OH)2. It occurs in nature as the mineral brucite. It is a white solid with low solubility in water (Ksp = 5.61×10−12). Magnesium hydroxide is a common component of antacids, such as milk of magnesia, as well as laxatives.How well is magnesium hydroxide absorbed?
Laxative action: Magnesium hydroxide produces its laxative effect by increasing the osmotic gradient in the gut and drawing in water, causing distention that stimulates peristalsis and bowel evacuation. Absorption: About 15% to 30% may be absorbed systemically (posing a potential risk to patients with renal failure).What drug class is magnesium hydroxide?
Magnesium hydroxide is in a class of medications called saline laxatives. It works by causing water to be retained with the stool. This increases the number of bowel movements and softens the stool so it is easier to pass.What acid is added to magnesium hydroxide to make magnesium chloride?
hydrochloric acid
Why is magnesium hydroxide insoluble in water?
Magnesium hydroxide appears to be insoluble in water. However, if it is shaken in water and filtered, the solution is slightly basic. This indicates that there are more hydroxide ions in solution than there were in the original water. This is because some magnesium hydroxide has dissolved.