How do you find the lattice constant for FCC?
How do you find the lattice constant for FCC?
If the space lattice is FCC, the lattice constant is given by the formula [4 x r / (2)1/2] and if the space lattice is BCC, then the lattice constant is given by the formula a = [4 x r / (3)1/2].
What is lattice constant for FCC crystal?
List of lattice constants
| Material | Lattice constant (Å) | Crystal structure |
|---|---|---|
| Mo | 3.142 | BCC |
| Pd | 3.859 | FCC |
| Ag | 4.079 | FCC |
| W | 3.155 | BCC |
What is the lattice structure of silver?
fcc crystal structure
Silver, Ag Silver has an fcc crystal structure with a basis of one silver atom. The lattice parameter, a, is 0.409 nm.
What is the lattice parameter for FCC?
0.3571 nm
The lattice parameter is 0.3571 nm for FCC iron and 0.2866 nm for BCC iron.
What is the relation between lattice constant and density?
The mass densities of crystalline materials are related to their crystal structures: (a) The lattice constant for crystalline Si is a = 5.43 Å, i.e., 5.43•10-10 m. Prove that its mass density is 2.33 gr/cm3. (b)Gem diamonds consist of pure carbon in the face-centered cubic (fcc) crystal structure.
What is the possible number of different types of lattice 3d?
In three dimensions, the lattices are categorized into seven crystal lattice “systems”. Within several of these, lattices supporting non-primitive unit cells can be defined. The classification scheme yields a total of 14 possible lattices (called Bravais lattices).
What is silver used for today?
Today silver is invaluable to solder and brazing alloys, batteries, dentistry, glass coatings, LED chips, medicine, nuclear reactors, photography, photovoltaic (or solar) energy, RFID chips (for tracking parcels or shipments worldwide), semiconductors, touch screens, water purification, wood preservatives and many …
What is the use of lattice parameters?
For example, knowledge of the lattice parameters can provide information on the thermal properties of a material, an indirect method to determine the compositions in a solid solution, a measure of the strain state, or an analysis of the defect structure.
How is the lattice length of a fcc crystal determined?
Using the hard sphere model, which imagines each atom as a discrete sphere, the FCC crystal has each atom touch along the face diagonal of the cube. That means that the face diagonal has a length of , so with a bit of geometry we find that the lattice parameter , or side length of the cube, has a length of .
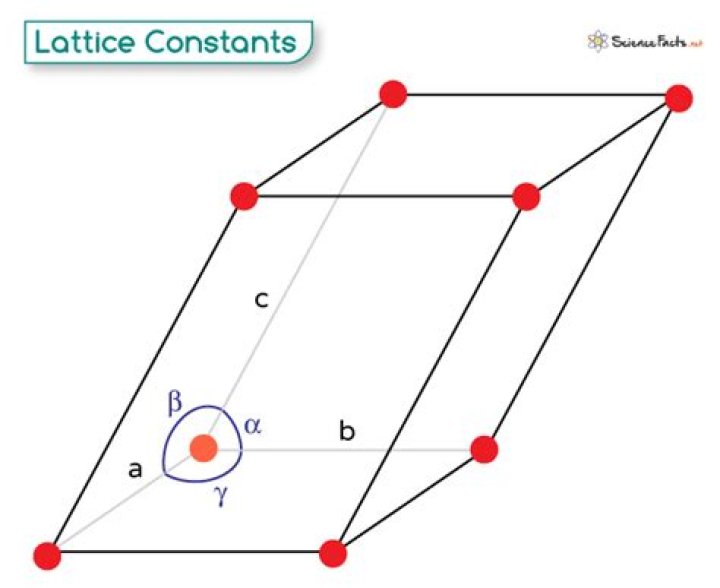
What is the lattice constant in a crystal?
A lattice constant describes the spacing between adjacent unit cells in a crystal structure. The unit cells or building blocks of the crystal are three dimensional and have three linear constants that describe the cell dimensions.
What are the constants of a face centered cubic lattice?
Face-Centered Cubic Lattice Constants The face-centered cubic lattice is a cube with an atom on each corner and each face. Using the hard sphere model, which imagines each atom as a discrete sphere, the FCC crystal has each atom touch along the face diagonal of the cube.
How is the atomic radius of copper determined?
The atomic radius of copper is 0.128 nm, while that of iron is 0.124 nm. Calculate the lattice constant, a, of the cubic unit cell. If the space lattice is SC, the lattice constant is given by the formula a = [2 x r].
How to calculate the lattice constant of copper?
The atomic radius of copper is 0.128 nm, while that of iron is 0.124 nm. Calculate the lattice constant, a, of the cubic unit cell. If the space lattice is SC, the lattice constant is given by the formula a = [2 x r]. For example, the lattice constant of the SC-crystallized polonium is [2 x 0.167 nm], or 0.334 nm.
How is the lattice constant used in cubic crystal systems?
A hard-sphere model is adopted, which allows you to visualize atoms in the cells as solid spheres. For cubic crystal systems, all three linear parameters are identical, so a single lattice constant is used to describe a cubic unit cell.
Which is the correct formula for the lattice constant v?
For orthorhombic, tetragonal and cubic lattices with β = 90° as well, then V = a b c . {\\displaystyle V=abc.} Matching of lattice structures between two different semiconductor materials allows a region of band gap change to be formed in a material without introducing a change in crystal structure.
Which is the formula for the lattice constant of polonium?
For example, the lattice constant of the SC-crystallized polonium is [2 x 0.167 nm], or 0.334 nm. If the space lattice is FCC, the lattice constant is given by the formula [4 x r / (2)1/2] and if the space lattice is BCC, then the lattice constant is given by the formula a = [4 x r / (3)1/2].

