How do you find the dipole moment in chemistry?
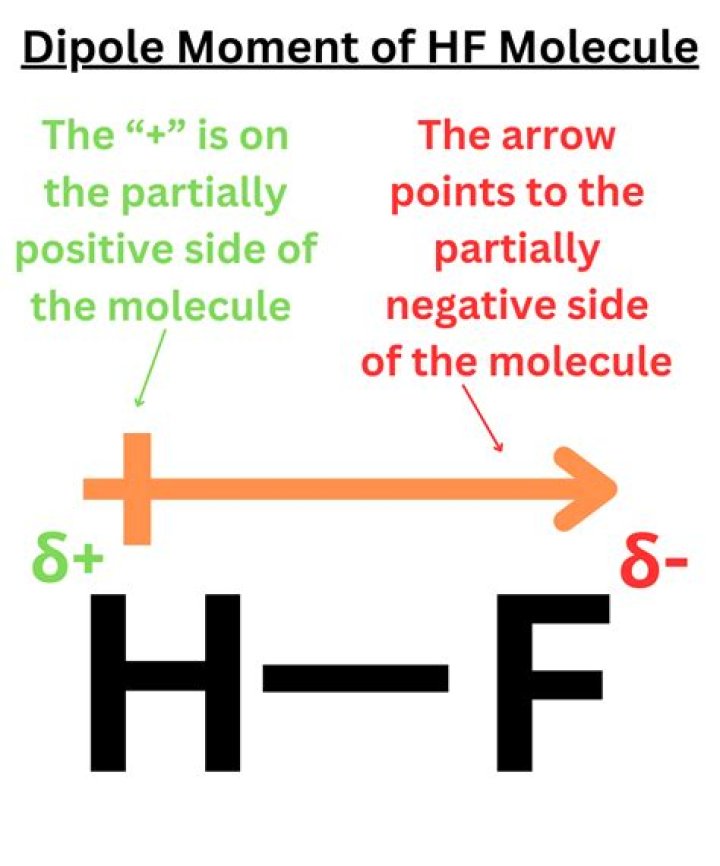
.
Then, how do you find the dipole moment?
The dipole moment is calculated by multiplying the distance between the hydrogen and oxygen atoms by the difference in their charge. Then, the angle between the atoms is used to find the net dipole moment. The angle formed by a water molecule is known to be 104.5° and the bond moment of the O-H bond is -1.5D.
One may also ask, what is a zero dipole moment? Zero dipole moment is the dipole moment between two atoms being zero. It depends on the polarities of individual bonds and the geometry of the atoms. For example CO2 , two opposing oxygen atom with carbon in the center as it's geometry . The dipole bonds are equal in magnitude but opposite in nature.
Accordingly, what is the dipole moment in chemistry?
A dipole moment is a quantity that describes two opposite charges separated by a distance. It is a quantity that we can measure for a molecule in the lab and thereby determine the size of the partial charges on the molecule (if we know the bond length).
What is dipole moment formula?
Dipole Moment Formula It is denoted by the Greek letter 'µ'. Mathematically, Dipole Moment (µ) = Charge (Q) * distance of separation (r) It is measured in Debye units denoted by 'D'. 1 D = 3.33564 × 10-30 C.m, where C is Coulomb and m denotes a meter.
Related Question AnswersWhat is the SI unit of dipole moment?
The SI unit for electric dipole moment is the coulomb-meter. This is too large to be practical on the molecular scale. Bond dipole moments are commonly measured in debyes, represented by the symbol D, which is obtained by measuring the charge in units of 10−10 statcoulomb and the distance d in Angstroms.Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.What do you mean by dipole?
In chemistry, a dipole usually refers to the separation of charges within a molecule between two covalently bonded atoms or atoms that share an ionic bond. For example, a water molecule (H2O) is a dipole.Why is dipole moment important?
Dipole moments tell us, on average, where the electrons in a molecule are. They can also tell us the shape of molecules. Also, molecular dipole moments are very important for many properties, such as ability to dissolve solutes, melting and boiling points, and reactivity in general.What is dipole moment example?
Polar molecules exhibit a large difference in electrical charge (a positive end and a negative end), otherwise known as a dipole moment. For example, ammonia (NHsub3) is a polar molecule. As you can see, ammonia consists of one nitrogen atom covalently bonded to three hydrogen atoms.Does water have a dipole moment?
Even though the total charge on a molecule is zero, the nature of chemical bonds is such that the positive and negative charges do not completely overlap in most molecules. Such molecules are said to be polar because they possess a permanent dipole moment. A good example is the dipole moment of the water molecule.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.What is dipole moment symbol?
μWhat is the dipole moment of HCl?
The dipole moment of HCl is 1.03 D, and the bond length is 127 pm.What is a dipole dipole force?
Dipole-dipole forces are attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. Dipole-dipole forces have strengths that range from 5 kJ to 20 kJ per mole. Polar molecules have a partial negative end and a partial positive end.Is dipole moment a scalar or vector?
Electric dipole moment is defined as the product of the magnitude of one of the charges and the distance between them. It is a a vector quantity . its direction is from the negative charge to the positive charge.Which has the largest dipole moment?
The larger the difference in electronegativities of bonded atoms, the larger the dipole moment. For example, NaCl has the highest dipole moment because it has an ionic bond (i.e. highest charge separation).What is resultant dipole moment?
In such molecules the dipole moment of the molecule is the vector sum of the dipole moments of various bonds. Due to the linear geometry the dipole moment of one C = O bond cancels that of another. Therefore, the resultant dipole moment of the molecule is zero and it is a non-polar molecule.Which is a dipole molecule?
Permanent dipoles. These occur when two atoms in a molecule have substantially different electronegativity: One atom attracts electrons more than another, becoming more negative, while the other atom becomes more positive. A molecule with a permanent dipole moment is called a polar molecule.Why is the dipole moment of bf3 zero?
Boron trifluoride is has zero dipole moment although it has three polar B—F bonds. This is because BF3 has sp2 hybridisation and regular trigonal planal geometry. And hence the individual dipole moments of polar bonds get cancelled and overall dipole moment is zero.What is permanent dipole moment?
Permanent dipoles: These occur when two atoms in a molecule have substantially different electronegativity: One atom attracts electrons more than another, becoming more negative, while the other atom becomes more positive. A molecule with a permanent dipole moment is called a polar molecule.Which molecules have a dipole moment?
Molecular Dipole Moment- carbon dioxide: 0 (despite having two polar C=O.
- carbon monoxide: 0.112 D.
- ozone: 0.53 D.
- phosgene: 1.17 D.
- water vapor: 1.85 D.
- hydrogen cyanide: 2.98 D.
- cyanamide: 4.27 D.
- potassium bromide: 10.41 D.

