How do you calculate buffering capacity?
How do you calculate buffering capacity?
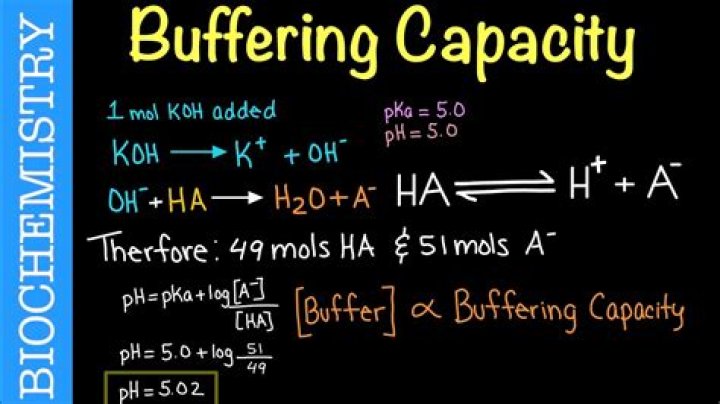
Use the buffer capacity equation to calculate the buffer capacity….pH = pKa + log10([A-]/[HA]) , where:
- [A-] is the concentration of a base in the buffer.
- [HA] is the concentration of a acid in the buffer.
- pKa is the dissociation constant of acid.
Which buffer has a higher buffering capacity?
Thus, the buffering capacity is influenced by the concentration of the weak acid and conjugate salt. So, a 1 molar acetate buffer has a higher buffering capacity than a 0.1 molar acetate buffer.
Is sodium citrate a buffer?
Citrate buffers can be used for RNA isolation, due to its ability to prevent base hydrolysis….Citrate Buffer (pH 3.0 to 6.2) Preparation and Recipe.
| Component | Amount | Concentration |
|---|---|---|
| Sodium Citrate dihydrate (mw: 294.10 g/mol) | 25.703 g | 0.0874 M |
| Citric Acid (mw: 192.12 g/mol) | 2.421 g | 0.0126 M |
Which solution has the best buffering capacity?
Buffer solutions with a pH equal to the pKa value of the acid (used to make this solution) have the greatest buffering capacity.
What is buffering capacity in chemistry?
The buffer capacity is defined as the amount of acid or base you can add without changing the pH by more than 1 pH unit.
What is buffering capacity of soil?
The buffering capacity of a soil indicates the capacity of the soil to resist pH change. When hydrogen ions in the soil solution are neutralised by lime, hydrogen ions from the soil surfaces are release into the soil solution to maintain equilibrium and resist increase in pH.
At what pH is the buffer capacity at its maximum?
pH = 7
Buffers have maximum buffering capacity at pH = 7.
What increases buffer capacity?
Buffering capacity refers to the amount of added acid or added base that can be neutralized by a buffer. It is determined by the concentrations of the conjugate acid and conjugate base. Buffering capacity increases as these concentrations increase.
What are citrate buffers?
Citrate is a buffer, or a chemical that’s used to help maintain the stability of medications, explains James Wu, PharmD, a pharmacist and manager of a Walgreens community pharmacy in Chicago. “It’s commonly found in injectable medications such as Humira to help it keep its chemical and physical properties,” he says.
How do you make sodium citrate buffer?
Citrate Buffer (0.1 M, pH 6.0) Preparation and Recipe
- Prepare 800 mL of distilled water in a suitable container.
- Add 24.269 g of Sodium Citrate dihydrate to the solution.
- Add 3.358 g of Citric Acid to the solution.
- Adjust solution to desired pH using 0.1N HCl (typically pH ≈ 6.0).
What increases buffering capacity?
Which solution has the greatest buffering capacity NH3?
The conjugate base-weak acid ratio for both Option B and Option E are close to 1, but the concentration of NH3 and NH4+ in Option E are higher. Therefore, Option E will have the greatest buffering capacity.

