How did Democritus find the atom
Democritus experiment was he took a simple seashell and break it in half. … To initially find the atom, Democritus conducted a simple experiment that can still be done today. Democritus built his work off of the theory of Leucipuss’. Democritus and his mentor Leucippus discovered the first atom theory.
How did Democritus do his experiment?
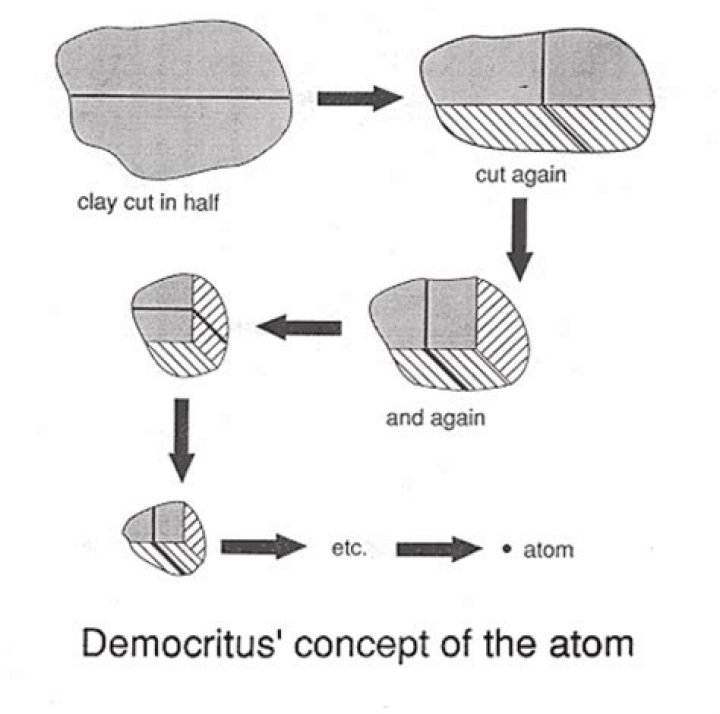
Democritus had a thought experiment. The idea was if you took a material and divided it half, you would have a smaller but identical chunk. If you keep dividing your material, there should eventually be a point where you’ve reached the smallest representative element of your material. That element is the”atom”.
How did Democritus find this new information what experiments did Democritus do?
Democritus experiment was he took a simple seashell and break it in half. … To initially find the atom, Democritus conducted a simple experiment that can still be done today. Democritus built his work off of the theory of Leucipuss’. Democritus and his mentor Leucippus discovered the first atom theory.
How did Democritus define the atom?
The theory of Democritus held that everything is composed of “atoms,” which are physically, but not geometrically, indivisible; that between atoms, there lies empty space; that atoms are indestructible, and have always been and always will be in motion; that there is an infinite number of atoms and of kinds of atoms, …When did Democritus discover the atom?
Around 400 B.C.E., the Greek philosopher Democritus introduced the idea of the atom as the basic building block matter.
What new information did Democritus contribute to the atom?
Democritus was a central figure in the development of the atomic theory of the universe. He theorized that all material bodies are made up of indivisibly small “atoms.” Aristotle famously rejected atomism in On Generation and Corruption.
How did Democritus come to his conclusions about the atom?
Democritus knew that if a stone was divided in half, the two halves would have essentially the same properties as the whole. Therefore, he reasoned that if the stone were to be continually cut into smaller and smaller pieces then; at some point, there would be a piece which would be so small as to be indivisible.
How did leucippus discover the atom?
Using scientific reasoning and observation, the Pre-Socratic philosopher Leucippus developed the theory of atomism, which posited that the whole universe is made up of solid atoms constantly moving through void, or empty space. Leucippus’ theory of atomism was developed even further by his pupil, Democritus.Why Democritus could not test his ideas about the atom Why do you think we are able to study the atom in the modern world?
Explain why Democritus was unable to experimentally verify his ideas. Democritus was unable to explain his ideas because he didn’t have the resources he need to prove the existence of atoms. … Aristotle believed that there was no empty space, therefore atoms could not move through empty space.
How did Dalton discover the atom?In 1803 Dalton discovered that oxygen combined with either one or two volumes of nitric oxide in closed vessels over water and this pioneering observation of integral multiple proportions provided important experimental evidence for his incipient atomic ideas.
Article first time published onWho was the first to discover atom?
Although the concept of the atom dates back to the ideas of Democritus, the English meteorologist and chemist John Dalton formulated the first modern description of it as the fundamental building block of chemical structures.
Why did Aristotle reject Democritus idea of the atom?
Aristotle: He rejected Democritus atomic theroy and didn’t think atoms move through empty spaces because he didn’t think that empty spaces existed. He believed that all matter consisted of four elements. Earth, Air, Water, and Fire.
What was JJ Thomson atomic theory?
Summary. J.J. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.”
How was Democritus atom different from Dalton's model of the atom?
The key difference between Democritus and Dalton atomic theory is that the Democritus atomic theory is an ancient theory that scientists later refined and elaborated whereas Dalton atomic theory is a comparatively modern, scientific theory that we cannot discard due its important statements.
What did Democritus and Leucippus discover?
In the 5th century BCE, Leucippus and his pupil Democritus proposed that all matter was composed of small indivisible particles called atoms. Nothing whatsoever is known about Leucippus except that he was the teacher of Democritus.
What did Leucippus introduced?
Leucippus, (flourished 5th century bc, probably at Miletus, on the west coast of Asia Minor), Greek philosopher credited by Aristotle and by Theophrastus with having originated the theory of atomism. It has been difficult to distinguish his contribution from that of his most famous pupil, Democritus.
Was Leucippus the first to propose an atomic theory?
The first proponents of an atomic theory were the Greek philosophers Leucippus and Democritus who proposed the following model in the fifth century B.C. 1.
Did Chadwick discover the neutron?
In 1932, Chadwick made a fundamental discovery in the domain of nuclear science: he proved the existence of neutrons – elementary particles devoid of any electrical charge. … For this epoch-making discovery he was awarded the Hughes Medal of the Royal Society in 1932, and subsequently the Nobel Prize for Physics in 1935.
What is Ernest Rutherford atomic theory?
Ernest Rutherford found that the atom is mostly empty space, with nearly all of its mass concentrated in a tiny central nucleus. The nucleus is positively charged and surrounded at a great distance by the negatively charged electrons.
What did Bohr discover about the atom?
The Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons. Bohr was the first to discover that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element.
Who discovered atom BYJU's?
According to the textbook that we read in our schools the man credited by modern science for conducting extensive research work on atoms and developing atomic theory goes to an English chemist and physicist John Dalton.
How much empty space is in an atom?
Recent studies indicate that protons have a radius of about . 84 × 10-15 meters, giving them a volume of about 2.5 × 10-45 cubic meters. A hydrogen atom is about 99.9999999999996% empty space.
Who proved atoms exist?
Einstein also in 1905 mathematically proved the existence of atoms, and thus helped revolutionize all the sciences through the use of statistics and probability. Atomic theory says that any liquid is made up of molecules (invisible in 1905).
How did Thomson discover the electron?
Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. He demonstrated that cathode rays were negatively charged. … Thomson realized that the accepted model of an atom did not account for negatively or positively charged particles.
Who discovered the proton?
It is 100 years since Ernest Rutherford published his results proving the existence of the proton. For decades, the proton was considered an elementary particle.
How did the concept of the atom change from Democritus to John Dalton?
Early History of the Atom Democritus proposed that different types and combinations of these particles were responsible for the various forms of matter. … Dalton’s atomic theory contained the following ideas: All atoms of a given element are identical. The atoms of different elements vary in mass and size.
Do you agree with Democritus that atoms are small hard particles?
He called this particle an atom. The word atom is from the Greek word atomos, meaning “not able to be divided.” Democritus said that all atoms are small, hard particles. He thought that atoms were made of a single material formed into different shapes and sizes. that led to the current atomic theory.