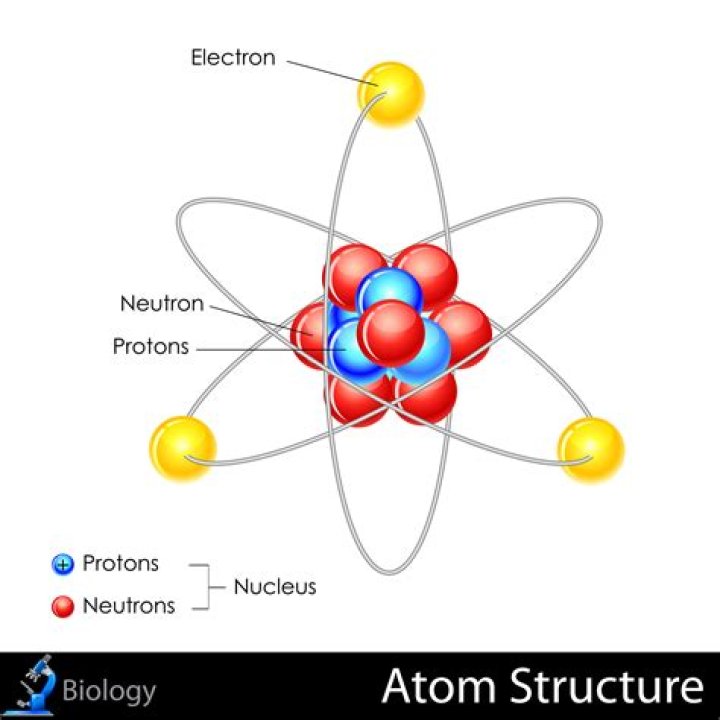
Originally Answered: How are atoms arranged in an element? Atoms are made up of electrons, protons and neutrons. The number of electrons and protons are always equal in an atom. So elements are determined by the numbers of electrons and protons in an atom..
Similarly one may ask, how are atoms in metals arranged?
1 Answer. Ella · Ernest Z. In a metallic solid atoms are arranged in a lattice-like 3D structure where there is a regular array of metal cations surrounded by a sea of delocalised electrons. The atoms have lots of shells of electrons surrounding the nucleus.
Furthermore, how are copper atoms arranged? Copper: metal whose atoms are arranged in closely packed hexagonal layers. First and fourth layers are in line with each other. This gives what is known as a face centred cubic pattern or cubic close packing. Copper is is a very good conductor of electricity and is commonly used in electrical circuits.
In this way, how are electrons arranged in an atom?
Electron Shells Here, electrons are arranged in energy levels, or shells, around the nucleus of an atom. Electrons that are in the first energy level (energy level 1) are closest to the nucleus and will have the lowest energy. Electrons further away from the nucleus will have higher energy.
How are atoms arranged in a solid?
Solids can hold their shape because their molecules are tightly packed together. Atoms and molecules in liquids and gases are bouncing and floating around, free to move where they want. The molecules in a solid are stuck in a specific structure or arrangement of atoms.
Related Question Answers
How strong are metallic bonds?
A metallic bond is the sharing of many detached electrons between many positive ions, where the electrons act as a "glue" giving the substance a definite structure. It is unlike covalent or ionic bonding. Metals have low ionization energy. Therefore, the valence electrons can be delocalized throughout the metals.Is Iron an alloy?
Iron is a great building material but steel (an alloy made by adding small amounts of nonmetallic carbon to iron) is stronger, harder, and rustproof. Aluminum is a very light metal but it's also very soft in its pure form.What are typical metals?
Typical metals These are the alkali metals, the alkaline earths, and aluminum. They have the following characteristics: They have an electronic structure similar to that of the inert gases with one, two, or three electrons in the outermost shell.How is metallic bond formed?
Metallic bonds form between metal atoms due to the transfer of valence electrons (outer electrons) from one metal atom to the other- these commonly form regions of electron density in what are called s and p orbitals (don't worry about these, they're just interesting).Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.Why is metallic bonding strong?
Metallic bonding The electrons from the outer shells of the metal atoms are delocalised , and are free to move through the whole structure. This sharing of delocalised electrons results in strong metallic bonding .What is metallic structure?
A metallic structure is primarily a crystalline structure consisting of closely packed atoms arranged in an orderly fashion.1,2. From: Engineering Textiles, 2009.What is 2n2 rule?
The sum of number of protons and number of electrons present in an atom is called atomic weight. 2n2 rule: This rule of arrangement of electrons according to the shell is known 2n2 rule where n means number of shell. For example: There is one proton in the nucleus of hydrogen atom, which moves in K shell path.What do you mean by Valency?
In chemistry, the valence or valency of an element is a measure of its combining power with other atoms when it forms chemical compounds or molecules. The concept of valence was developed in the second half of the 19th century and helped successfully explain the molecular structure of inorganic and organic compounds.How do you ionize an atom?
Ionization is the process by which ions are formed by gain or loss of an electron from an atom or molecule. If an atom or molecule gains an electron, it becomes negatively charged (an anion), and if it loses an electron, it becomes positively charged (a cation). Energy may be lost or gained in the formation of an ion.Whats is an atom?
An atom a fundamental piece of matter. An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud.What is Hund rule?
Hund's Rule. Hund's rule: every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals have the same spin.How many electrons are in each shell?
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.What is Shell in atom?
An electron shell is the outside part of an atom around the atomic nucleus. It is where the electrons are, and is a group of atomic orbitals with the same value of the principal quantum number n. Electron shells have one or more electron subshells, or sublevels.What is structure of atoms?
Atoms consist of three basic particles: protons, electrons, and neutrons. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).Why do all unpaired electrons need to have the same spin?
Electrons are negatively charged and, as a result, they repel each other. Electrons tend to minimize repulsion by occupying their own orbitals, rather than sharing an orbital with another electron. For the second rule, unpaired electrons in singly occupied orbitals have the same spins.How many atoms are in HCP?
6 atoms
Is FCC or BCC stronger?
FCC metals tend to have more slip systems, i.e. slip directions and slip planes than BCC. Thus FCC metals deform easier than BCC metals and thus they are more ductile. BCC metals are infact stronger than FCC metals.Is copper an atom or molecule?
You cannot ask if copper is an element or a molecule. into pieces, then the pieces would no longer be copper. The smallest unit of the element copper is 1 copper atom. not an element.