Does solubility of borax increase with temperature
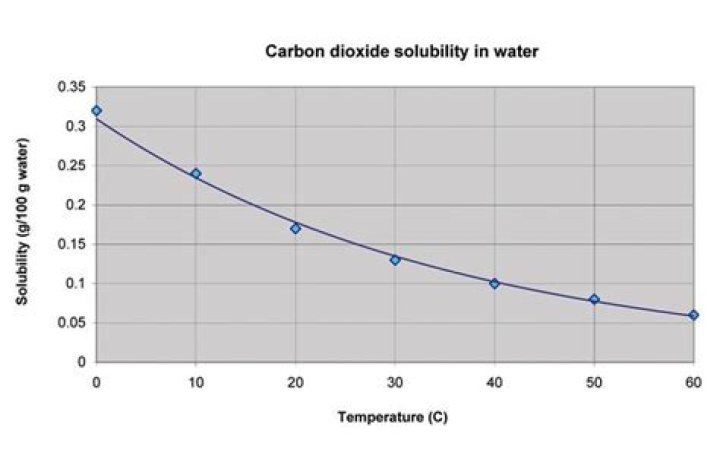
– The solubility of borax in water increases as temperature increases. – More borax is dissolved at higher temperatures, indicating heat is a reactant. … Thus the solvation of borax in water is endothermic.
Does the solubility of borax increase or decrease as temperature increases explain your answer?
Do you expect the solubility of Borax to increase or decrease as temperature increases? Select the option that best explains why. A. Solubility will increase, because as Tincreases the term becomes smaller therefore will get larger.
What temp does borax dissolve?
NamesDensity2.4 g/cm3 (anhydrous, solid) 1.73 g/cm3 (decahydrate, solid)Melting point743 °C (1,369 °F; 1,016 K) (anhydrous) 75 °C (decahydrate, decomposes)Boiling point1,575 °C (2,867 °F; 1,848 K) (anhydrous)Solubility in water31.7 g/L (both)
Is the dissolution of borax in water a temperature dependent?
The dissolution of borax in water is an endothermic reaction so it is a temperature-dependent reaction.Is the dissolution of borax an endothermic or exothermic process?
The dissolution (enthalpy or heat of solution) for borax (sodium tetraborate) is an endothermic process.
Is borax a salt?
Borax, or sodium borate, is an alkaline mineral salt with a powdery white appearance. Borax is most famous as a laundry washing powder.
Is borax dissolving in water a chemical change?
Chemical Change – Making Slime The borax should be dissolved in water to saturation (until no more borax will dissolve).
Why does borax not dissolve in water?
Hot water molecules are moving very fast and are spread way out which makes space available for more borax to dissolve into it. As the mixture cools, the water molecules slow down and move closer together. That means there’s less room for the dissolved borax and it begins to fall out of the water.What happens to borax in cold water?
The solubility of most solids increases with temperature. In other words, more Borax may be dissolved in hot water than cold water. So if a hot, saturated mixture is cooled, there’s more Borax than can be contained by the colder water, and so Borax may fall out of the mixture, forming crystals.
What happens when borax is dissolved in water?When borax reacts or it is dissolved in water, orthoboric acid, more commonly known as Boric acid (H₃BO₃) is formed along with sodium hydroxide (NaOH). The chemical reaction showing this change is done as shown below.
Article first time published onWhat is Delta G in chemistry?
Every chemical reaction involves a change in free energy, called delta G (∆G). The change in free energy can be calculated for any system that undergoes a change, such as a chemical reaction. To calculate ∆G, subtract the amount of energy lost to entropy (denoted as ∆S) from the total energy change of the system.
What does Delta H mean in thermodynamics?
Enthalpy changes Enthalpy change is the name given to the amount of heat evolved or absorbed in a reaction carried out at constant pressure. It is given the symbol ΔH, read as “delta H”.
Is borax crystals a chemical change?
As decahydrate borax crystals lose water from their chemical structure, there is a physical change in appearance from translucent to opaque. The hard translucent crystals begins to develop white powder on their surfaces.
What is difference between physical and chemical changes?
Physical changes only change the appearance of a substance, not its chemical composition. Chemical changes cause a substance to change into an entirely substance with a new chemical formula. Chemical changes are also known as chemical reactions.
When milk sours is it a chemical change?
Therefore milk souring is known as a chemical transition or chemical change because it ends up forming a new product that is the lactic acid, hence leaving the milk sour.
What is the pH of borax?
Borax is the sodium tetraborate decahydrate (Na2B4O7 · 10H2O) that, when dissolved in water, is hydrolyzed to boric acid and OH− anions, yielding a pH of about 9.13.
Why is borax banned in UK?
Borax is banned as a food additive here in the UK. … Borax is not available to buy in the UK or EU at all, due to the ‘borate’ group of chemicals being reclassified in 2010 as potentially hazardous to health.
Is borax a degreaser?
Borax is great at removing dried grease and food from aluminum or porcelain cookware because of its high pH level. The alkaline that helps make borax’s acidity breaks down tough grease and stains because it’s so strong.
Can you use borax with cold water?
Treat powdered borax the same way as dry baking soda. Sprinkle 1/2 cup borax into the empty washer tub before adding the dirty laundry. Be sure there are no lumps in the borax powder if you are using cold water because they may not dissolve properly. Borax works as a water softener and deodorizer.
Can you make borax crystals with cold water?
Hot water holds more borax crystals than cold water. That’s because heated water molecules move farther apart, making room for more of the borax crystals to dissolve.
Why do borax crystals grow better in cold?
Re: Borax Crystals Since heating up the water increases the amount of borax that can be dissolved into the water, cooling it down to a low temperature will decrease the amount and thus, the borax will fall out of the water and create more crystals.
How do you dissolve hardened borax?
If there is any hardened borax leftover from the experiment, boil water and pour it into the container that was used for the experiment. Using a stirring rod (ex: plastic spoon/fork) and stir to dissolve the remaining borax. Pour the waste away when it’s a bit cooler and rinse the container to get rid of any toxins.
Does borax melt plastic?
To best observe crystal growth on the strings it is preferable to use a glass container. … If you do use a plastic jar/container make sure that it can withstand the heat of boiling water and will not melt/deform. If any of your strands fuse together, gently pull them apart. Borax crystals are pretty hearty.
How do you make borax spray?
To create an all-purpose spray, dissolve 2 teaspoons of borax into 4 cups of hot water, then mix with 1 teaspoon of dish soap and 4 tablespoons of vinegar. Use this to clean countertops, appliances, windows, and more.
Does borax produce alkaline solution in water?
Borax solution is an alkaline solution due to presence of strong base sodium hydroxide which completely masks the acidic effect of weak acid boric acid and makes the property of solution alkaline.
What happens when you mix glue and borax?
Borax in water forms an ion called the borate ion. When the borax solution is added to the glue solution, the borate ions help link the long polymer molecules to each other so they cannot move and flow as easily.
When borax is heated strongly what does it give?
When borax is heated strongly it loses water molecules and swells. Then it is converted into a transparent liquid, solidifying which will form a glass-like material also called borax bead.
Is negative delta G favorable?
Re: Negative Delta G Reactions with a negative delta G are very spontaneous, and therefore highly favorable! The more favorable a reaction is, the more it will proceed towards the products.
Is negative delta G spontaneous?
Reactions with a negative ∆G release energy, which means that they can proceed without an energy input (are spontaneous). In contrast, reactions with a positive ∆G need an input of energy in order to take place (are non-spontaneous).
What affects Gibbs free energy?
The three critical factors in calculating the Gibbs free energy are enthalpy, entropy, and temperature.
What does ΔH 298 mean?
3O2(g)⟶2O3(g)ΔH∘298=+286kJ. Solution. ΔH∘f Δ H f ∘ is the enthalpy change for the formation of one mole of a substance in its standard state from the elements in their standard states. Thus, ΔH∘f Δ H f ∘ for O3(g) is the enthalpy change for the reaction: 32O2(g)⟶O3(g)