Does sodium react with liquid ammonia?
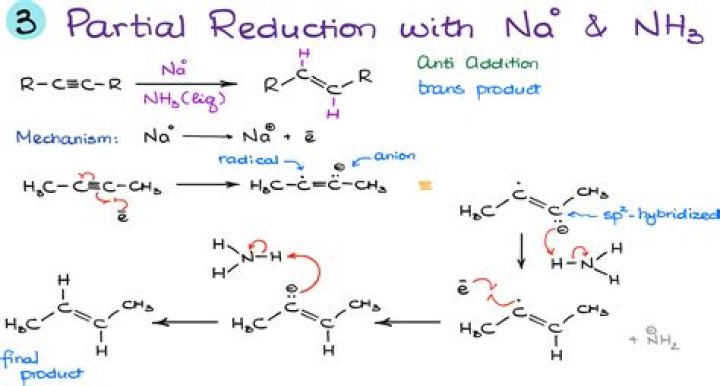
Does sodium react with liquid ammonia?
Sodium reacts with ammonia and forms sodium amide, that is, $NaN{H_2}$ and hydrogen gas is released.
What is the nature of NaNH2 in ammonia solvent?
In ammonia, NaNH2 forms conductive solutions, consistent with the presence of Na(NH3)6+ and NH2− ions.
What are the uses of Sodamide?
Sodium amide is used in the industrial production of indigo, hydrazine, and sodium cyanide. It is the reagent of choice for the drying of ammonia (liquid or gaseous) and is also widely used as a strong base in organic chemistry, often in liquid ammonia solution.
What is the formula of Sodamide?
NaNH2
Sodium amide/Formula
What happens when give chemical equation I sodium is reacted with liquid ammonia?
Note: Sodium like other alkali metals reacts with the liquid ammonia to produce deep blue colour solution. The blue solution of the sodium metal in ammonia is due to the formation of ammoniated electrons and ammoniated cations in the solution.
What happens when sodium reacts with ammonia?
-Sodium and ammonia react in the room temperature in a slow reactivity range to produce amide (sodium amide), with the liberation of hydrogen. – The reaction involving sodium and ammonia which results in formation of sodium amide and hydrogen can be accelerated by adding many metal oxides or metal.
Why solution become good conductor when sodium is added in liquid ammonia?
When sodium metal is added to ammonia, some of the sodium dissolves. Each of the dissolving sodium atoms loses an electron and becomes a cation. The solvated electrons give the blue color to the solution. Because of the mobility of the electrons, the solution is a good electrical conductor.
Does NaNH2 acts as an acid or base in liquid ammonia?
Sodium amide would act as a base in liquid ammonia because it would dissociate to produce the amide species, . This amide ion have tendency to accept proton, therefore it is a base.
What does NaNH2 do in a reaction?
As a strong base, NaNH2 will deprotonate alkynes, alcohols, and a host of other functional groups with acidic protons such as esters and ketones. As a base, it’s often used in situations where a strong, small base is required.
What is the meaning of Sodamide?
sodamide. / (ˈsəʊdəˌmaɪd) / noun. a white crystalline compound used as a dehydrating agent, as a chemical reagent, and in making sodium cyanide.
What is the formula of sodium ethoxide?
C2H5ONa
Sodium ethoxide/Formula