Do CH bonds have high potential energy?
Do CH bonds have high potential energy?
Explanation: Chemical bonds certainly “contain” potential energy, and the atoms want to move to a lower potential energy (become more stable). When methane, CH4 , forms, the valence electrons end up in more stable (lower energy) C-H bonds. These bonds are fairly strong, so methane is relatively inert.
Does gases have potential energy?
Ideal gases are a very simple system of noninteracting particles. The only energy involved is the kinetic energy of the gas particles. There is no potential energy.
Do chemical bonds have potential energy?
Energy, potential energy, is stored in the covalent bonds holding atoms together in the form of molecules. This is often called chemical energy. Some kinetic energy may be exchanged, but the total energy will stay the same.
What is a pairwise potential?
The pairwise potential energy, V(r) , between two non-bonded atoms can be expressed as a function of internuclear separation, r , as follows, A revised set of parameters has been calculated, which use the same Van der Waals radius of a given atom for all pairwise distances, no matter what the other atom.
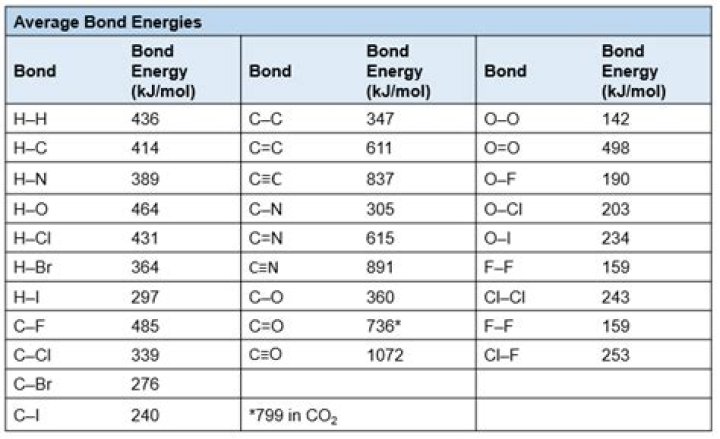
Is potential energy stored in CH and CC bonds?
free energy is immediately reabsorbed to form the new covalent bonds that hold the hydrogen atoms to the oxygen atoms in the new water molecule….
| Bond | Energy (kcal/mole) |
|---|---|
| O-O | 58 |
| O=O | 116 |
| C-H | 98 |
| C-O | 78 |
Does ATP or ADP have more potential energy?
ATP has more potential energy than ADP since it has three negatively charged phosphates held together vs only two in ADP.
Do ideal gases have kinetic energy?
An ideal gas is defined as one in which all collisions between atoms or molecules are perfectly eleastic and in which there are no intermolecular attractive forces. In such a gas, all the internal energy is in the form of kinetic energy and any change in internal energy is accompanied by a change in temperature.
Do real gases have kinetic energy?
Gas particles are in constant rapid motion in random directions . The fast motion of gas particles gives them a relatively large amount of kinetic energy.
Is chemical energy potential or kinetic?
Chemical energy is potential energy. Kinetic energy is the energy of motion. Chemical energy deals with the energy stored in the bonds between atoms….
Is a chemical bonds kinetic or potential energy?
Chemical energy, the energy stored in chemical bonds, is thus considered a form of potential energy.
What is Epsilon in Lennard Jones potential?
ϵ is the well depth and a measure of how strongly the two particles attract each other. σ is the distance at which the intermolecular potential between the two particles is zero (Figure 1). σ gives a measurement of how close two nonbonding particles can get and is thus referred to as the van der Waals radius.
What does potential mean in statistics?
Statistical potentials are Gibbs-energy functions derived from the frequency of occurrence of conformations in a database of structures (Shortle, 2003).