Are p orbitals sp2?
Are p orbitals sp2?
The atomic s- and p-orbitals in boron’s outer shell mix to form three equivalent hybrid orbitals. These particular orbitals are called sp2 hybrids, meaning that this set of orbitals derives from one s- orbital and two p-orbitals of the free atom. The Lewis structure for etheneThe carbon atoms are sp2 hybridized.
What is the P sp2 hybridization?
The sp2 hybridization is the mixing of one s and two p atomic orbitals, which involves the promotion of one electron in the s orbital to one of the 2p atomic orbitals. The new set of formed hybrid orbitals creates trigonal structures, creating a molecular geometry of 120 degrees.
What is the hybridization of P in P4O10?
If P4O10 is formed, it is sp3 hybridized and it is a form of tetrahedral.
What is hybridization of so2?
In sulphur dioxide, the hybridization that takes place is sp2 type. To determine this, we will first look at the sulphur atom which will be the central atom. During the formation of SO2, this central atom is bonded with two oxygen atoms and their structure can be represented as O=S=O.
How many p orbitals are in sp3?
three p orbitals
In sp³ hybridization, one s orbital and three p orbitals hybridize to form four sp³ orbitals, each consisting of 25% s character and 75% p character. This type of hybridization is required whenever an atom is surrounded by four groups of electrons.
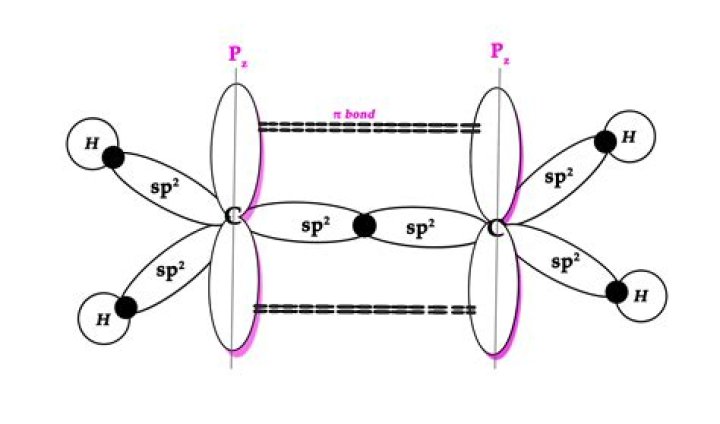
Why alkene is sp2 hybridized?
Alkenes. Each sp2 hybridized carbon forms three σ bonds using three sp2 hybridized orbitals. The π bond is weaker than the σ bond, but is strong enough to prevent rotation of the C=C bond. Therefore, alkenes are planar, with each carbon being trigonal planar.
What is hybridization of p in PCl5?
In PCl5, the hybridisation of P atom is sp3d.
How many number of p − O − p bonds is P4O10?
Hint: Hint: In ${P_4}{O_{10}}$, there are six bonds between phosphorus-oxygen -phosphorus and every phosphorus has double bond with oxygen that is there are four such bonds where oxygen is $s{p^2}$ hybridized.
What is hybridization of SO3?
The hybridization in SO3 is sp2 because of the formation of one sigma bond and one pi bond.

